
Peer Recognized
Make a name in academia

How to Write a Research Paper: the LEAP approach (+cheat sheet)
In this article I will show you how to write a research paper using the four LEAP writing steps. The LEAP academic writing approach is a step-by-step method for turning research results into a published paper .
The LEAP writing approach has been the cornerstone of the 70 + research papers that I have authored and the 3700+ citations these paper have accumulated within 9 years since the completion of my PhD. I hope the LEAP approach will help you just as much as it has helped me to make an real, tangible impact with my research.
What is the LEAP research paper writing approach?
I designed the LEAP writing approach not only for merely writing the papers. My goal with the writing system was to show young scientists how to first think about research results and then how to efficiently write each section of the research paper.
In other words, you will see how to write a research paper by first analyzing the results and then building a logical, persuasive arguments. In this way, instead of being afraid of writing research paper, you will be able to rely on the paper writing process to help you with what is the most demanding task in getting published – thinking.
The four research paper writing steps according to the LEAP approach:

I will show each of these steps in detail. And you will be able to download the LEAP cheat sheet for using with every paper you write.
But before I tell you how to efficiently write a research paper, I want to show you what is the problem with the way scientists typically write a research paper and why the LEAP approach is more efficient.
How scientists typically write a research paper (and why it isn’t efficient)
Writing a research paper can be tough, especially for a young scientist. Your reasoning needs to be persuasive and thorough enough to convince readers of your arguments. The description has to be derived from research evidence, from prior art, and from your own judgment. This is a tough feat to accomplish.
The figure below shows the sequence of the different parts of a typical research paper. Depending on the scientific journal, some sections might be merged or nonexistent, but the general outline of a research paper will remain very similar.

Here is the problem: Most people make the mistake of writing in this same sequence.
While the structure of scientific articles is designed to help the reader follow the research, it does little to help the scientist write the paper. This is because the layout of research articles starts with the broad (introduction) and narrows down to the specifics (results). See in the figure below how the research paper is structured in terms of the breath of information that each section entails.
How to write a research paper according to the LEAP approach
For a scientist, it is much easier to start writing a research paper with laying out the facts in the narrow sections (i.e. results), step back to describe them (i.e. write the discussion), and step back again to explain the broader picture in the introduction.
For example, it might feel intimidating to start writing a research paper by explaining your research’s global significance in the introduction, while it is easy to plot the figures in the results. When plotting the results, there is not much room for wiggle: the results are what they are.
Starting to write a research papers from the results is also more fun because you finally get to see and understand the complete picture of the research that you have worked on.
Most importantly, following the LEAP approach will help you first make sense of the results yourself and then clearly communicate them to the readers. That is because the sequence of writing allows you to slowly understand the meaning of the results and then develop arguments for presenting to your readers.
I have personally been able to write and submit a research article in three short days using this method.
Step 1: Lay Out the Facts

You have worked long hours on a research project that has produced results and are no doubt curious to determine what they exactly mean. There is no better way to do this than by preparing figures, graphics and tables. This is what the first LEAP step is focused on – diving into the results.
How to p repare charts and tables for a research paper
Your first task is to try out different ways of visually demonstrating the research results. In many fields, the central items of a journal paper will be charts that are based on the data generated during research. In other fields, these might be conceptual diagrams, microscopy images, schematics and a number of other types of scientific graphics which should visually communicate the research study and its results to the readers. If you have reasonably small number of data points, data tables might be useful as well.
Tips for preparing charts and tables
- Try multiple chart types but in the finished paper only use the one that best conveys the message you want to present to the readers
- Follow the eight chart design progressions for selecting and refining a data chart for your paper: https://peerrecognized.com/chart-progressions
- Prepare scientific graphics and visualizations for your paper using the scientific graphic design cheat sheet: https://peerrecognized.com/tools-for-creating-scientific-illustrations/
How to describe the results of your research
Now that you have your data charts, graphics and tables laid out in front of you – describe what you see in them. Seek to answer the question: What have I found? Your statements should progress in a logical sequence and be backed by the visual information. Since, at this point, you are simply explaining what everyone should be able to see for themselves, you can use a declarative tone: The figure X demonstrates that…
Tips for describing the research results :
- Answer the question: “ What have I found? “
- Use declarative tone since you are simply describing observations
Step 2: Explain the results

The core aspect of your research paper is not actually the results; it is the explanation of their meaning. In the second LEAP step, you will do some heavy lifting by guiding the readers through the results using logic backed by previous scientific research.
How to define the Message of a research paper
To define the central message of your research paper, imagine how you would explain your research to a colleague in 20 seconds . If you succeed in effectively communicating your paper’s message, a reader should be able to recount your findings in a similarly concise way even a year after reading it. This clarity will increase the chances that someone uses the knowledge you generated, which in turn raises the likelihood of citations to your research paper.
Tips for defining the paper’s central message :
- Write the paper’s core message in a single sentence or two bullet points
- Write the core message in the header of the research paper manuscript
How to write the Discussion section of a research paper
In the discussion section you have to demonstrate why your research paper is worthy of publishing. In other words, you must now answer the all-important So what? question . How well you do so will ultimately define the success of your research paper.
Here are three steps to get started with writing the discussion section:
- Write bullet points of the things that convey the central message of the research article (these may evolve into subheadings later on).
- Make a list with the arguments or observations that support each idea.
- Finally, expand on each point to make full sentences and paragraphs.
Tips for writing the discussion section:
- What is the meaning of the results?
- Was the hypothesis confirmed?
- Write bullet points that support the core message
- List logical arguments for each bullet point, group them into sections
- Instead of repeating research timeline, use a presentation sequence that best supports your logic
- Convert arguments to full paragraphs; be confident but do not overhype
- Refer to both supportive and contradicting research papers for maximum credibility
How to write the Conclusions of a research paper
Since some readers might just skim through your research paper and turn directly to the conclusions, it is a good idea to make conclusion a standalone piece. In the first few sentences of the conclusions, briefly summarize the methodology and try to avoid using abbreviations (if you do, explain what they mean).
After this introduction, summarize the findings from the discussion section. Either paragraph style or bullet-point style conclusions can be used. I prefer the bullet-point style because it clearly separates the different conclusions and provides an easy-to-digest overview for the casual browser. It also forces me to be more succinct.
Tips for writing the conclusion section :
- Summarize the key findings, starting with the most important one
- Make conclusions standalone (short summary, avoid abbreviations)
- Add an optional take-home message and suggest future research in the last paragraph
How to refine the Objective of a research paper
The objective is a short, clear statement defining the paper’s research goals. It can be included either in the final paragraph of the introduction, or as a separate subsection after the introduction. Avoid writing long paragraphs with in-depth reasoning, references, and explanation of methodology since these belong in other sections. The paper’s objective can often be written in a single crisp sentence.
Tips for writing the objective section :
- The objective should ask the question that is answered by the central message of the research paper
- The research objective should be clear long before writing a paper. At this point, you are simply refining it to make sure it is addressed in the body of the paper.
How to write the Methodology section of your research paper
When writing the methodology section, aim for a depth of explanation that will allow readers to reproduce the study . This means that if you are using a novel method, you will have to describe it thoroughly. If, on the other hand, you applied a standardized method, or used an approach from another paper, it will be enough to briefly describe it with reference to the detailed original source.
Remember to also detail the research population, mention how you ensured representative sampling, and elaborate on what statistical methods you used to analyze the results.
Tips for writing the methodology section :
- Include enough detail to allow reproducing the research
- Provide references if the methods are known
- Create a methodology flow chart to add clarity
- Describe the research population, sampling methodology, statistical methods for result analysis
- Describe what methodology, test methods, materials, and sample groups were used in the research.
Step 3: Advertize the research
Step 3 of the LEAP writing approach is designed to entice the casual browser into reading your research paper. This advertising can be done with an informative title, an intriguing abstract, as well as a thorough explanation of the underlying need for doing the research within the introduction.

How to write the Introduction of a research paper
The introduction section should leave no doubt in the mind of the reader that what you are doing is important and that this work could push scientific knowledge forward. To do this convincingly, you will need to have a good knowledge of what is state-of-the-art in your field. You also need be able to see the bigger picture in order to demonstrate the potential impacts of your research work.
Think of the introduction as a funnel, going from wide to narrow, as shown in the figure below:
- Start with a brief context to explain what do we already know,
- Follow with the motivation for the research study and explain why should we care about it,
- Explain the research gap you are going to bridge within this research paper,
- Describe the approach you will take to solve the problem.
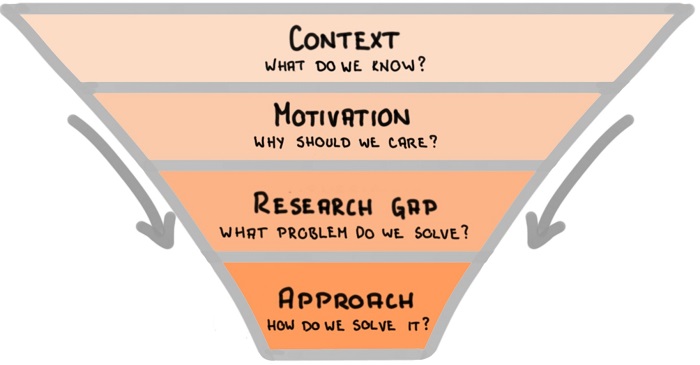
Tips for writing the introduction section :
- Follow the Context – Motivation – Research gap – Approach funnel for writing the introduction
- Explain how others tried and how you plan to solve the research problem
- Do a thorough literature review before writing the introduction
- Start writing the introduction by using your own words, then add references from the literature
How to prepare the Abstract of a research paper
The abstract acts as your paper’s elevator pitch and is therefore best written only after the main text is finished. In this one short paragraph you must convince someone to take on the time-consuming task of reading your whole research article. So, make the paper easy to read, intriguing, and self-explanatory; avoid jargon and abbreviations.
How to structure the abstract of a research paper:
- The abstract is a single paragraph that follows this structure:
- Problem: why did we research this
- Methodology: typically starts with the words “Here we…” that signal the start of own contribution.
- Results: what we found from the research.
- Conclusions: show why are the findings important
How to compose a research paper Title
The title is the ultimate summary of a research paper. It must therefore entice someone looking for information to click on a link to it and continue reading the article. A title is also used for indexing purposes in scientific databases, so a representative and optimized title will play large role in determining if your research paper appears in search results at all.
Tips for coming up with a research paper title:
- Capture curiosity of potential readers using a clear and descriptive title
- Include broad terms that are often searched
- Add details that uniquely identify the researched subject of your research paper
- Avoid jargon and abbreviations
- Use keywords as title extension (instead of duplicating the words) to increase the chance of appearing in search results
How to prepare Highlights and Graphical Abstract
Highlights are three to five short bullet-point style statements that convey the core findings of the research paper. Notice that the focus is on the findings, not on the process of getting there.
A graphical abstract placed next to the textual abstract visually summarizes the entire research paper in a single, easy-to-follow figure. I show how to create a graphical abstract in my book Research Data Visualization and Scientific Graphics.
Tips for preparing highlights and graphical abstract:
- In highlights show core findings of the research paper (instead of what you did in the study).
- In graphical abstract show take-home message or methodology of the research paper. Learn more about creating a graphical abstract in this article.
Step 4: Prepare for submission

Sometimes it seems that nuclear fusion will stop on the star closest to us (read: the sun will stop to shine) before a submitted manuscript is published in a scientific journal. The publication process routinely takes a long time, and after submitting the manuscript you have very little control over what happens. To increase the chances of a quick publication, you must do your homework before submitting the manuscript. In the fourth LEAP step, you make sure that your research paper is published in the most appropriate journal as quickly and painlessly as possible.
How to select a scientific Journal for your research paper
The best way to find a journal for your research paper is it to review which journals you used while preparing your manuscript. This source listing should provide some assurance that your own research paper, once published, will be among similar articles and, thus, among your field’s trusted sources.
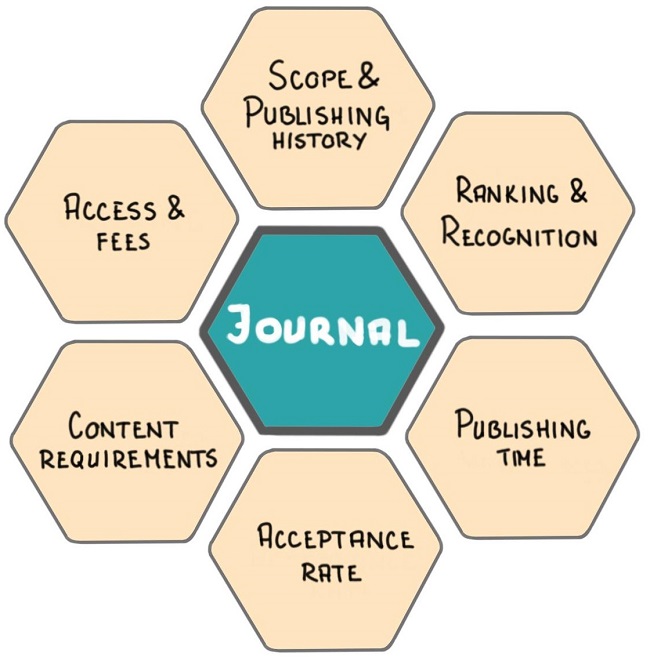
After this initial selection of hand-full of scientific journals, consider the following six parameters for selecting the most appropriate journal for your research paper (read this article to review each step in detail):
- Scope and publishing history
- Ranking and Recognition
- Publishing time
- Acceptance rate
- Content requirements
- Access and Fees
How to select a journal for your research paper:
- Use the six parameters to select the most appropriate scientific journal for your research paper
- Use the following tools for journal selection: https://peerrecognized.com/journals
- Follow the journal’s “Authors guide” formatting requirements
How to Edit you manuscript
No one can write a finished research paper on their first attempt. Before submitting, make sure to take a break from your work for a couple of days, or even weeks. Try not to think about the manuscript during this time. Once it has faded from your memory, it is time to return and edit. The pause will allow you to read the manuscript from a fresh perspective and make edits as necessary.
I have summarized the most useful research paper editing tools in this article.
Tips for editing a research paper:
- Take time away from the research paper to forget about it; then returning to edit,
- Start by editing the content: structure, headings, paragraphs, logic, figures
- Continue by editing the grammar and language; perform a thorough language check using academic writing tools
- Read the entire paper out loud and correct what sounds weird
How to write a compelling Cover Letter for your paper
Begin the cover letter by stating the paper’s title and the type of paper you are submitting (review paper, research paper, short communication). Next, concisely explain why your study was performed, what was done, and what the key findings are. State why the results are important and what impact they might have in the field. Make sure you mention how your approach and findings relate to the scope of the journal in order to show why the article would be of interest to the journal’s readers.
I wrote a separate article that explains what to include in a cover letter here. You can also download a cover letter template from the article.
Tips for writing a cover letter:
- Explain how the findings of your research relate to journal’s scope
- Tell what impact the research results will have
- Show why the research paper will interest the journal’s audience
- Add any legal statements as required in journal’s guide for authors
How to Answer the Reviewers
Reviewers will often ask for new experiments, extended discussion, additional details on the experimental setup, and so forth. In principle, your primary winning tactic will be to agree with the reviewers and follow their suggestions whenever possible. After all, you must earn their blessing in order to get your paper published.
Be sure to answer each review query and stick to the point. In the response to the reviewers document write exactly where in the paper you have made any changes. In the paper itself, highlight the changes using a different color. This way the reviewers are less likely to re-read the entire article and suggest new edits.
In cases when you don’t agree with the reviewers, it makes sense to answer more thoroughly. Reviewers are scientifically minded people and so, with enough logical and supported argument, they will eventually be willing to see things your way.
Tips for answering the reviewers:
- Agree with most review comments, but if you don’t, thoroughly explain why
- Highlight changes in the manuscript
- Do not take the comments personally and cool down before answering
The LEAP research paper writing cheat sheet
Imagine that you are back in grad school and preparing to take an exam on the topic: “How to write a research paper”. As an exemplary student, you would, most naturally, create a cheat sheet summarizing the subject… Well, I did it for you.
This one-page summary of the LEAP research paper writing technique will remind you of the key research paper writing steps. Print it out and stick it to a wall in your office so that you can review it whenever you are writing a new research paper.

Now that we have gone through the four LEAP research paper writing steps, I hope you have a good idea of how to write a research paper. It can be an enjoyable process and once you get the hang of it, the four LEAP writing steps should even help you think about and interpret the research results. This process should enable you to write a well-structured, concise, and compelling research paper.
Have fund with writing your next research paper. I hope it will turn out great!
Learn writing papers that get cited
The LEAP writing approach is a blueprint for writing research papers. But to be efficient and write papers that get cited, you need more than that.
My name is Martins Zaumanis and in my interactive course Research Paper Writing Masterclass I will show you how to visualize your research results, frame a message that convinces your readers, and write each section of the paper. Step-by-step.
And of course – you will learn to respond the infamous Reviewer No.2.

Hey! My name is Martins Zaumanis and I am a materials scientist in Switzerland ( Google Scholar ). As the first person in my family with a PhD, I have first-hand experience of the challenges starting scientists face in academia. With this blog, I want to help young researchers succeed in academia. I call the blog “Peer Recognized”, because peer recognition is what lifts academic careers and pushes science forward.
Besides this blog, I have written the Peer Recognized book series and created the Peer Recognized Academy offering interactive online courses.
Related articles:
One comment
- Pingback: Research Paper Outline with Key Sentence Skeleton (+Paper Template)
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
I want to join the Peer Recognized newsletter!
This site uses Akismet to reduce spam. Learn how your comment data is processed .
Privacy Overview
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
Copyright © 2024 Martins Zaumanis
Contacts: [email protected]
Privacy Policy
- Privacy Policy

Home » Research Paper – Structure, Examples and Writing Guide
Research Paper – Structure, Examples and Writing Guide
Table of Contents

Research Paper
Definition:
Research Paper is a written document that presents the author’s original research, analysis, and interpretation of a specific topic or issue.
It is typically based on Empirical Evidence, and may involve qualitative or quantitative research methods, or a combination of both. The purpose of a research paper is to contribute new knowledge or insights to a particular field of study, and to demonstrate the author’s understanding of the existing literature and theories related to the topic.
Structure of Research Paper
The structure of a research paper typically follows a standard format, consisting of several sections that convey specific information about the research study. The following is a detailed explanation of the structure of a research paper:
The title page contains the title of the paper, the name(s) of the author(s), and the affiliation(s) of the author(s). It also includes the date of submission and possibly, the name of the journal or conference where the paper is to be published.
The abstract is a brief summary of the research paper, typically ranging from 100 to 250 words. It should include the research question, the methods used, the key findings, and the implications of the results. The abstract should be written in a concise and clear manner to allow readers to quickly grasp the essence of the research.
Introduction
The introduction section of a research paper provides background information about the research problem, the research question, and the research objectives. It also outlines the significance of the research, the research gap that it aims to fill, and the approach taken to address the research question. Finally, the introduction section ends with a clear statement of the research hypothesis or research question.
Literature Review
The literature review section of a research paper provides an overview of the existing literature on the topic of study. It includes a critical analysis and synthesis of the literature, highlighting the key concepts, themes, and debates. The literature review should also demonstrate the research gap and how the current study seeks to address it.
The methods section of a research paper describes the research design, the sample selection, the data collection and analysis procedures, and the statistical methods used to analyze the data. This section should provide sufficient detail for other researchers to replicate the study.
The results section presents the findings of the research, using tables, graphs, and figures to illustrate the data. The findings should be presented in a clear and concise manner, with reference to the research question and hypothesis.
The discussion section of a research paper interprets the findings and discusses their implications for the research question, the literature review, and the field of study. It should also address the limitations of the study and suggest future research directions.
The conclusion section summarizes the main findings of the study, restates the research question and hypothesis, and provides a final reflection on the significance of the research.
The references section provides a list of all the sources cited in the paper, following a specific citation style such as APA, MLA or Chicago.
How to Write Research Paper
You can write Research Paper by the following guide:
- Choose a Topic: The first step is to select a topic that interests you and is relevant to your field of study. Brainstorm ideas and narrow down to a research question that is specific and researchable.
- Conduct a Literature Review: The literature review helps you identify the gap in the existing research and provides a basis for your research question. It also helps you to develop a theoretical framework and research hypothesis.
- Develop a Thesis Statement : The thesis statement is the main argument of your research paper. It should be clear, concise and specific to your research question.
- Plan your Research: Develop a research plan that outlines the methods, data sources, and data analysis procedures. This will help you to collect and analyze data effectively.
- Collect and Analyze Data: Collect data using various methods such as surveys, interviews, observations, or experiments. Analyze data using statistical tools or other qualitative methods.
- Organize your Paper : Organize your paper into sections such as Introduction, Literature Review, Methods, Results, Discussion, and Conclusion. Ensure that each section is coherent and follows a logical flow.
- Write your Paper : Start by writing the introduction, followed by the literature review, methods, results, discussion, and conclusion. Ensure that your writing is clear, concise, and follows the required formatting and citation styles.
- Edit and Proofread your Paper: Review your paper for grammar and spelling errors, and ensure that it is well-structured and easy to read. Ask someone else to review your paper to get feedback and suggestions for improvement.
- Cite your Sources: Ensure that you properly cite all sources used in your research paper. This is essential for giving credit to the original authors and avoiding plagiarism.
Research Paper Example
Note : The below example research paper is for illustrative purposes only and is not an actual research paper. Actual research papers may have different structures, contents, and formats depending on the field of study, research question, data collection and analysis methods, and other factors. Students should always consult with their professors or supervisors for specific guidelines and expectations for their research papers.
Research Paper Example sample for Students:
Title: The Impact of Social Media on Mental Health among Young Adults
Abstract: This study aims to investigate the impact of social media use on the mental health of young adults. A literature review was conducted to examine the existing research on the topic. A survey was then administered to 200 university students to collect data on their social media use, mental health status, and perceived impact of social media on their mental health. The results showed that social media use is positively associated with depression, anxiety, and stress. The study also found that social comparison, cyberbullying, and FOMO (Fear of Missing Out) are significant predictors of mental health problems among young adults.
Introduction: Social media has become an integral part of modern life, particularly among young adults. While social media has many benefits, including increased communication and social connectivity, it has also been associated with negative outcomes, such as addiction, cyberbullying, and mental health problems. This study aims to investigate the impact of social media use on the mental health of young adults.
Literature Review: The literature review highlights the existing research on the impact of social media use on mental health. The review shows that social media use is associated with depression, anxiety, stress, and other mental health problems. The review also identifies the factors that contribute to the negative impact of social media, including social comparison, cyberbullying, and FOMO.
Methods : A survey was administered to 200 university students to collect data on their social media use, mental health status, and perceived impact of social media on their mental health. The survey included questions on social media use, mental health status (measured using the DASS-21), and perceived impact of social media on their mental health. Data were analyzed using descriptive statistics and regression analysis.
Results : The results showed that social media use is positively associated with depression, anxiety, and stress. The study also found that social comparison, cyberbullying, and FOMO are significant predictors of mental health problems among young adults.
Discussion : The study’s findings suggest that social media use has a negative impact on the mental health of young adults. The study highlights the need for interventions that address the factors contributing to the negative impact of social media, such as social comparison, cyberbullying, and FOMO.
Conclusion : In conclusion, social media use has a significant impact on the mental health of young adults. The study’s findings underscore the need for interventions that promote healthy social media use and address the negative outcomes associated with social media use. Future research can explore the effectiveness of interventions aimed at reducing the negative impact of social media on mental health. Additionally, longitudinal studies can investigate the long-term effects of social media use on mental health.
Limitations : The study has some limitations, including the use of self-report measures and a cross-sectional design. The use of self-report measures may result in biased responses, and a cross-sectional design limits the ability to establish causality.
Implications: The study’s findings have implications for mental health professionals, educators, and policymakers. Mental health professionals can use the findings to develop interventions that address the negative impact of social media use on mental health. Educators can incorporate social media literacy into their curriculum to promote healthy social media use among young adults. Policymakers can use the findings to develop policies that protect young adults from the negative outcomes associated with social media use.
References :
- Twenge, J. M., & Campbell, W. K. (2019). Associations between screen time and lower psychological well-being among children and adolescents: Evidence from a population-based study. Preventive medicine reports, 15, 100918.
- Primack, B. A., Shensa, A., Escobar-Viera, C. G., Barrett, E. L., Sidani, J. E., Colditz, J. B., … & James, A. E. (2017). Use of multiple social media platforms and symptoms of depression and anxiety: A nationally-representative study among US young adults. Computers in Human Behavior, 69, 1-9.
- Van der Meer, T. G., & Verhoeven, J. W. (2017). Social media and its impact on academic performance of students. Journal of Information Technology Education: Research, 16, 383-398.
Appendix : The survey used in this study is provided below.
Social Media and Mental Health Survey
- How often do you use social media per day?
- Less than 30 minutes
- 30 minutes to 1 hour
- 1 to 2 hours
- 2 to 4 hours
- More than 4 hours
- Which social media platforms do you use?
- Others (Please specify)
- How often do you experience the following on social media?
- Social comparison (comparing yourself to others)
- Cyberbullying
- Fear of Missing Out (FOMO)
- Have you ever experienced any of the following mental health problems in the past month?
- Do you think social media use has a positive or negative impact on your mental health?
- Very positive
- Somewhat positive
- Somewhat negative
- Very negative
- In your opinion, which factors contribute to the negative impact of social media on mental health?
- Social comparison
- In your opinion, what interventions could be effective in reducing the negative impact of social media on mental health?
- Education on healthy social media use
- Counseling for mental health problems caused by social media
- Social media detox programs
- Regulation of social media use
Thank you for your participation!
Applications of Research Paper
Research papers have several applications in various fields, including:
- Advancing knowledge: Research papers contribute to the advancement of knowledge by generating new insights, theories, and findings that can inform future research and practice. They help to answer important questions, clarify existing knowledge, and identify areas that require further investigation.
- Informing policy: Research papers can inform policy decisions by providing evidence-based recommendations for policymakers. They can help to identify gaps in current policies, evaluate the effectiveness of interventions, and inform the development of new policies and regulations.
- Improving practice: Research papers can improve practice by providing evidence-based guidance for professionals in various fields, including medicine, education, business, and psychology. They can inform the development of best practices, guidelines, and standards of care that can improve outcomes for individuals and organizations.
- Educating students : Research papers are often used as teaching tools in universities and colleges to educate students about research methods, data analysis, and academic writing. They help students to develop critical thinking skills, research skills, and communication skills that are essential for success in many careers.
- Fostering collaboration: Research papers can foster collaboration among researchers, practitioners, and policymakers by providing a platform for sharing knowledge and ideas. They can facilitate interdisciplinary collaborations and partnerships that can lead to innovative solutions to complex problems.
When to Write Research Paper
Research papers are typically written when a person has completed a research project or when they have conducted a study and have obtained data or findings that they want to share with the academic or professional community. Research papers are usually written in academic settings, such as universities, but they can also be written in professional settings, such as research organizations, government agencies, or private companies.
Here are some common situations where a person might need to write a research paper:
- For academic purposes: Students in universities and colleges are often required to write research papers as part of their coursework, particularly in the social sciences, natural sciences, and humanities. Writing research papers helps students to develop research skills, critical thinking skills, and academic writing skills.
- For publication: Researchers often write research papers to publish their findings in academic journals or to present their work at academic conferences. Publishing research papers is an important way to disseminate research findings to the academic community and to establish oneself as an expert in a particular field.
- To inform policy or practice : Researchers may write research papers to inform policy decisions or to improve practice in various fields. Research findings can be used to inform the development of policies, guidelines, and best practices that can improve outcomes for individuals and organizations.
- To share new insights or ideas: Researchers may write research papers to share new insights or ideas with the academic or professional community. They may present new theories, propose new research methods, or challenge existing paradigms in their field.
Purpose of Research Paper
The purpose of a research paper is to present the results of a study or investigation in a clear, concise, and structured manner. Research papers are written to communicate new knowledge, ideas, or findings to a specific audience, such as researchers, scholars, practitioners, or policymakers. The primary purposes of a research paper are:
- To contribute to the body of knowledge : Research papers aim to add new knowledge or insights to a particular field or discipline. They do this by reporting the results of empirical studies, reviewing and synthesizing existing literature, proposing new theories, or providing new perspectives on a topic.
- To inform or persuade: Research papers are written to inform or persuade the reader about a particular issue, topic, or phenomenon. They present evidence and arguments to support their claims and seek to persuade the reader of the validity of their findings or recommendations.
- To advance the field: Research papers seek to advance the field or discipline by identifying gaps in knowledge, proposing new research questions or approaches, or challenging existing assumptions or paradigms. They aim to contribute to ongoing debates and discussions within a field and to stimulate further research and inquiry.
- To demonstrate research skills: Research papers demonstrate the author’s research skills, including their ability to design and conduct a study, collect and analyze data, and interpret and communicate findings. They also demonstrate the author’s ability to critically evaluate existing literature, synthesize information from multiple sources, and write in a clear and structured manner.
Characteristics of Research Paper
Research papers have several characteristics that distinguish them from other forms of academic or professional writing. Here are some common characteristics of research papers:
- Evidence-based: Research papers are based on empirical evidence, which is collected through rigorous research methods such as experiments, surveys, observations, or interviews. They rely on objective data and facts to support their claims and conclusions.
- Structured and organized: Research papers have a clear and logical structure, with sections such as introduction, literature review, methods, results, discussion, and conclusion. They are organized in a way that helps the reader to follow the argument and understand the findings.
- Formal and objective: Research papers are written in a formal and objective tone, with an emphasis on clarity, precision, and accuracy. They avoid subjective language or personal opinions and instead rely on objective data and analysis to support their arguments.
- Citations and references: Research papers include citations and references to acknowledge the sources of information and ideas used in the paper. They use a specific citation style, such as APA, MLA, or Chicago, to ensure consistency and accuracy.
- Peer-reviewed: Research papers are often peer-reviewed, which means they are evaluated by other experts in the field before they are published. Peer-review ensures that the research is of high quality, meets ethical standards, and contributes to the advancement of knowledge in the field.
- Objective and unbiased: Research papers strive to be objective and unbiased in their presentation of the findings. They avoid personal biases or preconceptions and instead rely on the data and analysis to draw conclusions.
Advantages of Research Paper
Research papers have many advantages, both for the individual researcher and for the broader academic and professional community. Here are some advantages of research papers:
- Contribution to knowledge: Research papers contribute to the body of knowledge in a particular field or discipline. They add new information, insights, and perspectives to existing literature and help advance the understanding of a particular phenomenon or issue.
- Opportunity for intellectual growth: Research papers provide an opportunity for intellectual growth for the researcher. They require critical thinking, problem-solving, and creativity, which can help develop the researcher’s skills and knowledge.
- Career advancement: Research papers can help advance the researcher’s career by demonstrating their expertise and contributions to the field. They can also lead to new research opportunities, collaborations, and funding.
- Academic recognition: Research papers can lead to academic recognition in the form of awards, grants, or invitations to speak at conferences or events. They can also contribute to the researcher’s reputation and standing in the field.
- Impact on policy and practice: Research papers can have a significant impact on policy and practice. They can inform policy decisions, guide practice, and lead to changes in laws, regulations, or procedures.
- Advancement of society: Research papers can contribute to the advancement of society by addressing important issues, identifying solutions to problems, and promoting social justice and equality.
Limitations of Research Paper
Research papers also have some limitations that should be considered when interpreting their findings or implications. Here are some common limitations of research papers:
- Limited generalizability: Research findings may not be generalizable to other populations, settings, or contexts. Studies often use specific samples or conditions that may not reflect the broader population or real-world situations.
- Potential for bias : Research papers may be biased due to factors such as sample selection, measurement errors, or researcher biases. It is important to evaluate the quality of the research design and methods used to ensure that the findings are valid and reliable.
- Ethical concerns: Research papers may raise ethical concerns, such as the use of vulnerable populations or invasive procedures. Researchers must adhere to ethical guidelines and obtain informed consent from participants to ensure that the research is conducted in a responsible and respectful manner.
- Limitations of methodology: Research papers may be limited by the methodology used to collect and analyze data. For example, certain research methods may not capture the complexity or nuance of a particular phenomenon, or may not be appropriate for certain research questions.
- Publication bias: Research papers may be subject to publication bias, where positive or significant findings are more likely to be published than negative or non-significant findings. This can skew the overall findings of a particular area of research.
- Time and resource constraints: Research papers may be limited by time and resource constraints, which can affect the quality and scope of the research. Researchers may not have access to certain data or resources, or may be unable to conduct long-term studies due to practical limitations.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

Ethical Considerations – Types, Examples and...

Implications in Research – Types, Examples and...

Chapter Summary & Overview – Writing Guide...

Scope of the Research – Writing Guide and...

Background of The Study – Examples and Writing...

Institutional Review Board – Application Sample...
Instant insights, infinite possibilities
- How to write a research paper
Last updated
11 January 2024
Reviewed by
With proper planning, knowledge, and framework, completing a research paper can be a fulfilling and exciting experience.
Though it might initially sound slightly intimidating, this guide will help you embrace the challenge.
By documenting your findings, you can inspire others and make a difference in your field. Here's how you can make your research paper unique and comprehensive.
- What is a research paper?
Research papers allow you to demonstrate your knowledge and understanding of a particular topic. These papers are usually lengthier and more detailed than typical essays, requiring deeper insight into the chosen topic.
To write a research paper, you must first choose a topic that interests you and is relevant to the field of study. Once you’ve selected your topic, gathering as many relevant resources as possible, including books, scholarly articles, credible websites, and other academic materials, is essential. You must then read and analyze these sources, summarizing their key points and identifying gaps in the current research.
You can formulate your ideas and opinions once you thoroughly understand the existing research. To get there might involve conducting original research, gathering data, or analyzing existing data sets. It could also involve presenting an original argument or interpretation of the existing research.
Writing a successful research paper involves presenting your findings clearly and engagingly, which might involve using charts, graphs, or other visual aids to present your data and using concise language to explain your findings. You must also ensure your paper adheres to relevant academic formatting guidelines, including proper citations and references.
Overall, writing a research paper requires a significant amount of time, effort, and attention to detail. However, it is also an enriching experience that allows you to delve deeply into a subject that interests you and contribute to the existing body of knowledge in your chosen field.
- How long should a research paper be?
Research papers are deep dives into a topic. Therefore, they tend to be longer pieces of work than essays or opinion pieces.
However, a suitable length depends on the complexity of the topic and your level of expertise. For instance, are you a first-year college student or an experienced professional?
Also, remember that the best research papers provide valuable information for the benefit of others. Therefore, the quality of information matters most, not necessarily the length. Being concise is valuable.
Following these best practice steps will help keep your process simple and productive:
1. Gaining a deep understanding of any expectations
Before diving into your intended topic or beginning the research phase, take some time to orient yourself. Suppose there’s a specific topic assigned to you. In that case, it’s essential to deeply understand the question and organize your planning and approach in response. Pay attention to the key requirements and ensure you align your writing accordingly.
This preparation step entails
Deeply understanding the task or assignment
Being clear about the expected format and length
Familiarizing yourself with the citation and referencing requirements
Understanding any defined limits for your research contribution
Where applicable, speaking to your professor or research supervisor for further clarification
2. Choose your research topic
Select a research topic that aligns with both your interests and available resources. Ideally, focus on a field where you possess significant experience and analytical skills. In crafting your research paper, it's crucial to go beyond summarizing existing data and contribute fresh insights to the chosen area.
Consider narrowing your focus to a specific aspect of the topic. For example, if exploring the link between technology and mental health, delve into how social media use during the pandemic impacts the well-being of college students. Conducting interviews and surveys with students could provide firsthand data and unique perspectives, adding substantial value to the existing knowledge.
When finalizing your topic, adhere to legal and ethical norms in the relevant area (this ensures the integrity of your research, protects participants' rights, upholds intellectual property standards, and ensures transparency and accountability). Following these principles not only maintains the credibility of your work but also builds trust within your academic or professional community.
For instance, in writing about medical research, consider legal and ethical norms , including patient confidentiality laws and informed consent requirements. Similarly, if analyzing user data on social media platforms, be mindful of data privacy regulations, ensuring compliance with laws governing personal information collection and use. Aligning with legal and ethical standards not only avoids potential issues but also underscores the responsible conduct of your research.
3. Gather preliminary research
Once you’ve landed on your topic, it’s time to explore it further. You’ll want to discover more about available resources and existing research relevant to your assignment at this stage.
This exploratory phase is vital as you may discover issues with your original idea or realize you have insufficient resources to explore the topic effectively. This key bit of groundwork allows you to redirect your research topic in a different, more feasible, or more relevant direction if necessary.
Spending ample time at this stage ensures you gather everything you need, learn as much as you can about the topic, and discover gaps where the topic has yet to be sufficiently covered, offering an opportunity to research it further.
4. Define your research question
To produce a well-structured and focused paper, it is imperative to formulate a clear and precise research question that will guide your work. Your research question must be informed by the existing literature and tailored to the scope and objectives of your project. By refining your focus, you can produce a thoughtful and engaging paper that effectively communicates your ideas to your readers.
5. Write a thesis statement
A thesis statement is a one-to-two-sentence summary of your research paper's main argument or direction. It serves as an overall guide to summarize the overall intent of the research paper for you and anyone wanting to know more about the research.
A strong thesis statement is:
Concise and clear: Explain your case in simple sentences (avoid covering multiple ideas). It might help to think of this section as an elevator pitch.
Specific: Ensure that there is no ambiguity in your statement and that your summary covers the points argued in the paper.
Debatable: A thesis statement puts forward a specific argument––it is not merely a statement but a debatable point that can be analyzed and discussed.
Here are three thesis statement examples from different disciplines:
Psychology thesis example: "We're studying adults aged 25-40 to see if taking short breaks for mindfulness can help with stress. Our goal is to find practical ways to manage anxiety better."
Environmental science thesis example: "This research paper looks into how having more city parks might make the air cleaner and keep people healthier. I want to find out if more green spaces means breathing fewer carcinogens in big cities."
UX research thesis example: "This study focuses on improving mobile banking for older adults using ethnographic research, eye-tracking analysis, and interactive prototyping. We investigate the usefulness of eye-tracking analysis with older individuals, aiming to spark debate and offer fresh perspectives on UX design and digital inclusivity for the aging population."
6. Conduct in-depth research
A research paper doesn’t just include research that you’ve uncovered from other papers and studies but your fresh insights, too. You will seek to become an expert on your topic––understanding the nuances in the current leading theories. You will analyze existing research and add your thinking and discoveries. It's crucial to conduct well-designed research that is rigorous, robust, and based on reliable sources. Suppose a research paper lacks evidence or is biased. In that case, it won't benefit the academic community or the general public. Therefore, examining the topic thoroughly and furthering its understanding through high-quality research is essential. That usually means conducting new research. Depending on the area under investigation, you may conduct surveys, interviews, diary studies , or observational research to uncover new insights or bolster current claims.
7. Determine supporting evidence
Not every piece of research you’ve discovered will be relevant to your research paper. It’s important to categorize the most meaningful evidence to include alongside your discoveries. It's important to include evidence that doesn't support your claims to avoid exclusion bias and ensure a fair research paper.
8. Write a research paper outline
Before diving in and writing the whole paper, start with an outline. It will help you to see if more research is needed, and it will provide a framework by which to write a more compelling paper. Your supervisor may even request an outline to approve before beginning to write the first draft of the full paper. An outline will include your topic, thesis statement, key headings, short summaries of the research, and your arguments.
9. Write your first draft
Once you feel confident about your outline and sources, it’s time to write your first draft. While penning a long piece of content can be intimidating, if you’ve laid the groundwork, you will have a structure to help you move steadily through each section. To keep up motivation and inspiration, it’s often best to keep the pace quick. Stopping for long periods can interrupt your flow and make jumping back in harder than writing when things are fresh in your mind.
10. Cite your sources correctly
It's always a good practice to give credit where it's due, and the same goes for citing any works that have influenced your paper. Building your arguments on credible references adds value and authenticity to your research. In the formatting guidelines section, you’ll find an overview of different citation styles (MLA, CMOS, or APA), which will help you meet any publishing or academic requirements and strengthen your paper's credibility. It is essential to follow the guidelines provided by your school or the publication you are submitting to ensure the accuracy and relevance of your citations.
11. Ensure your work is original
It is crucial to ensure the originality of your paper, as plagiarism can lead to serious consequences. To avoid plagiarism, you should use proper paraphrasing and quoting techniques. Paraphrasing is rewriting a text in your own words while maintaining the original meaning. Quoting involves directly citing the source. Giving credit to the original author or source is essential whenever you borrow their ideas or words. You can also use plagiarism detection tools such as Scribbr or Grammarly to check the originality of your paper. These tools compare your draft writing to a vast database of online sources. If you find any accidental plagiarism, you should correct it immediately by rephrasing or citing the source.
12. Revise, edit, and proofread
One of the essential qualities of excellent writers is their ability to understand the importance of editing and proofreading. Even though it's tempting to call it a day once you've finished your writing, editing your work can significantly improve its quality. It's natural to overlook the weaker areas when you've just finished writing a paper. Therefore, it's best to take a break of a day or two, or even up to a week, to refresh your mind. This way, you can return to your work with a new perspective. After some breathing room, you can spot any inconsistencies, spelling and grammar errors, typos, or missing citations and correct them.
- The best research paper format
The format of your research paper should align with the requirements set forth by your college, school, or target publication.
There is no one “best” format, per se. Depending on the stated requirements, you may need to include the following elements:
Title page: The title page of a research paper typically includes the title, author's name, and institutional affiliation and may include additional information such as a course name or instructor's name.
Table of contents: Include a table of contents to make it easy for readers to find specific sections of your paper.
Abstract: The abstract is a summary of the purpose of the paper.
Methods : In this section, describe the research methods used. This may include collecting data , conducting interviews, or doing field research .
Results: Summarize the conclusions you drew from your research in this section.
Discussion: In this section, discuss the implications of your research . Be sure to mention any significant limitations to your approach and suggest areas for further research.
Tables, charts, and illustrations: Use tables, charts, and illustrations to help convey your research findings and make them easier to understand.
Works cited or reference page: Include a works cited or reference page to give credit to the sources that you used to conduct your research.
Bibliography: Provide a list of all the sources you consulted while conducting your research.
Dedication and acknowledgments : Optionally, you may include a dedication and acknowledgments section to thank individuals who helped you with your research.
- General style and formatting guidelines
Formatting your research paper means you can submit it to your college, journal, or other publications in compliance with their criteria.
Research papers tend to follow the American Psychological Association (APA), Modern Language Association (MLA), or Chicago Manual of Style (CMOS) guidelines.
Here’s how each style guide is typically used:
Chicago Manual of Style (CMOS):
CMOS is a versatile style guide used for various types of writing. It's known for its flexibility and use in the humanities. CMOS provides guidelines for citations, formatting, and overall writing style. It allows for both footnotes and in-text citations, giving writers options based on their preferences or publication requirements.
American Psychological Association (APA):
APA is common in the social sciences. It’s hailed for its clarity and emphasis on precision. It has specific rules for citing sources, creating references, and formatting papers. APA style uses in-text citations with an accompanying reference list. It's designed to convey information efficiently and is widely used in academic and scientific writing.
Modern Language Association (MLA):
MLA is widely used in the humanities, especially literature and language studies. It emphasizes the author-page format for in-text citations and provides guidelines for creating a "Works Cited" page. MLA is known for its focus on the author's name and the literary works cited. It’s frequently used in disciplines that prioritize literary analysis and critical thinking.
To confirm you're using the latest style guide, check the official website or publisher's site for updates, consult academic resources, and verify the guide's publication date. Online platforms and educational resources may also provide summaries and alerts about any revisions or additions to the style guide.
Citing sources
When working on your research paper, it's important to cite the sources you used properly. Your citation style will guide you through this process. Generally, there are three parts to citing sources in your research paper:
First, provide a brief citation in the body of your essay. This is also known as a parenthetical or in-text citation.
Second, include a full citation in the Reference list at the end of your paper. Different types of citations include in-text citations, footnotes, and reference lists.
In-text citations include the author's surname and the date of the citation.
Footnotes appear at the bottom of each page of your research paper. They may also be summarized within a reference list at the end of the paper.
A reference list includes all of the research used within the paper at the end of the document. It should include the author, date, paper title, and publisher listed in the order that aligns with your citation style.
10 research paper writing tips:
Following some best practices is essential to writing a research paper that contributes to your field of study and creates a positive impact.
These tactics will help you structure your argument effectively and ensure your work benefits others:
Clear and precise language: Ensure your language is unambiguous. Use academic language appropriately, but keep it simple. Also, provide clear takeaways for your audience.
Effective idea separation: Organize the vast amount of information and sources in your paper with paragraphs and titles. Create easily digestible sections for your readers to navigate through.
Compelling intro: Craft an engaging introduction that captures your reader's interest. Hook your audience and motivate them to continue reading.
Thorough revision and editing: Take the time to review and edit your paper comprehensively. Use tools like Grammarly to detect and correct small, overlooked errors.
Thesis precision: Develop a clear and concise thesis statement that guides your paper. Ensure that your thesis aligns with your research's overall purpose and contribution.
Logical flow of ideas: Maintain a logical progression throughout the paper. Use transitions effectively to connect different sections and maintain coherence.
Critical evaluation of sources: Evaluate and critically assess the relevance and reliability of your sources. Ensure that your research is based on credible and up-to-date information.
Thematic consistency: Maintain a consistent theme throughout the paper. Ensure that all sections contribute cohesively to the overall argument.
Relevant supporting evidence: Provide concise and relevant evidence to support your arguments. Avoid unnecessary details that may distract from the main points.
Embrace counterarguments: Acknowledge and address opposing views to strengthen your position. Show that you have considered alternative arguments in your field.
7 research tips
If you want your paper to not only be well-written but also contribute to the progress of human knowledge, consider these tips to take your paper to the next level:
Selecting the appropriate topic: The topic you select should align with your area of expertise, comply with the requirements of your project, and have sufficient resources for a comprehensive investigation.
Use academic databases: Academic databases such as PubMed, Google Scholar, and JSTOR offer a wealth of research papers that can help you discover everything you need to know about your chosen topic.
Critically evaluate sources: It is important not to accept research findings at face value. Instead, it is crucial to critically analyze the information to avoid jumping to conclusions or overlooking important details. A well-written research paper requires a critical analysis with thorough reasoning to support claims.
Diversify your sources: Expand your research horizons by exploring a variety of sources beyond the standard databases. Utilize books, conference proceedings, and interviews to gather diverse perspectives and enrich your understanding of the topic.
Take detailed notes: Detailed note-taking is crucial during research and can help you form the outline and body of your paper.
Stay up on trends: Keep abreast of the latest developments in your field by regularly checking for recent publications. Subscribe to newsletters, follow relevant journals, and attend conferences to stay informed about emerging trends and advancements.
Engage in peer review: Seek feedback from peers or mentors to ensure the rigor and validity of your research . Peer review helps identify potential weaknesses in your methodology and strengthens the overall credibility of your findings.
- The real-world impact of research papers
Writing a research paper is more than an academic or business exercise. The experience provides an opportunity to explore a subject in-depth, broaden one's understanding, and arrive at meaningful conclusions. With careful planning, dedication, and hard work, writing a research paper can be a fulfilling and enriching experience contributing to advancing knowledge.
How do I publish my research paper?
Many academics wish to publish their research papers. While challenging, your paper might get traction if it covers new and well-written information. To publish your research paper, find a target publication, thoroughly read their guidelines, format your paper accordingly, and send it to them per their instructions. You may need to include a cover letter, too. After submission, your paper may be peer-reviewed by experts to assess its legitimacy, quality, originality, and methodology. Following review, you will be informed by the publication whether they have accepted or rejected your paper.
What is a good opening sentence for a research paper?
Beginning your research paper with a compelling introduction can ensure readers are interested in going further. A relevant quote, a compelling statistic, or a bold argument can start the paper and hook your reader. Remember, though, that the most important aspect of a research paper is the quality of the information––not necessarily your ability to storytell, so ensure anything you write aligns with your goals.
Research paper vs. a research proposal—what’s the difference?
While some may confuse research papers and proposals, they are different documents.
A research proposal comes before a research paper. It is a detailed document that outlines an intended area of exploration. It includes the research topic, methodology, timeline, sources, and potential conclusions. Research proposals are often required when seeking approval to conduct research.
A research paper is a summary of research findings. A research paper follows a structured format to present those findings and construct an argument or conclusion.
Should you be using a customer insights hub?
Do you want to discover previous research faster?
Do you share your research findings with others?
Do you analyze research data?
Start for free today, add your research, and get to key insights faster
Editor’s picks
Last updated: 18 April 2023
Last updated: 27 February 2023
Last updated: 22 August 2024
Last updated: 5 February 2023
Last updated: 16 April 2023
Last updated: 9 March 2023
Last updated: 30 April 2024
Last updated: 12 December 2023
Last updated: 11 March 2024
Last updated: 4 July 2024
Last updated: 6 March 2024
Last updated: 5 March 2024
Last updated: 13 May 2024
Latest articles
Related topics, .css-je19u9{-webkit-align-items:flex-end;-webkit-box-align:flex-end;-ms-flex-align:flex-end;align-items:flex-end;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-flex-direction:row;-ms-flex-direction:row;flex-direction:row;-webkit-box-flex-wrap:wrap;-webkit-flex-wrap:wrap;-ms-flex-wrap:wrap;flex-wrap:wrap;-webkit-box-pack:center;-ms-flex-pack:center;-webkit-justify-content:center;justify-content:center;row-gap:0;text-align:center;max-width:671px;}@media (max-width: 1079px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}}@media (max-width: 799px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}} decide what to .css-1kiodld{max-height:56px;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-align-items:center;-webkit-box-align:center;-ms-flex-align:center;align-items:center;}@media (max-width: 1079px){.css-1kiodld{display:none;}} build next, decide what to build next.
- 10 research paper
Log in or sign up
Get started for free

Research Methods: A Student's Comprehensive Guide: Structure
- Research Approaches
- Types of Sources
- Accessing Resources
- Evaluating Sources
- Question Crafting
- Search Strategies
- Annotated Bibliography
- Literature Reviews
- Citations This link opens in a new window
Research Paper
Welcome to the art of crafting a research paper! Think of this as your roadmap to creating a well-structured and impactful study. We’ll walk you through each crucial component—from introducing your topic with flair to wrapping up with a strong conclusion. Whether you're diving into your first research project or polishing your latest masterpiece, this guide is here to make the journey smoother and more enjoyable. Get ready to turn your research into a compelling narrative that not only showcases your findings but also captivates your readers.
- Paper Snapshot
Introduction
Methodology, research paper structure: a snapshot.
Before diving into the individual components, let's take a quick look at the full structure of a research paper. This snapshot will help you visualize how each section fits together to form a cohesive and well-organized paper.
- Introduce your topic and research question.
- Provide background and context to set up your study.
- Summarize relevant existing research.
- Highlight key studies, theories, and gaps in the literature.
- Describe your research design and methods.
- Explain your data collection and analysis processes.
- Present your findings clearly.
- Use visuals, like charts and tables, to enhance understanding.
- Analyze and interpret the results.
- Discuss the broader implications of your findings and acknowledge limitations.
- Recap your key findings.
- Suggest areas for future research and offer final reflections.
With this snapshot, you now have a high-level view of the main components of your research paper. You can explore each section in detail in the following tabs.
The introduction serves as your reader's first impression of your paper. It should draw them in with a compelling overview of your topic, clearly outline your research question or thesis, and establish the importance of your study.
Key Components
Opening Statement
- Start strong with an attention-grabbing hook: a striking fact, thought-provoking quote, or an interesting anecdote that relates to your research.
Background Information
- Provide necessary context to help readers understand the relevance and scope of your study. You can include key historical information, theoretical context, or a brief overview of previous research.
Research Question or Thesis Statement
- This is the heart of your introduction. State your research question or thesis in a clear, concise manner, so readers know exactly what you are investigating.
Scope and Objectives
- Clearly define the boundaries of your research. What will your paper cover, and what will it not address? This helps frame your work for readers.
Significance of the Study
- Explain why your research matters. Does it fill a gap in existing research? Is it practically useful? Emphasize the value and contribution your paper brings to the field.
Tips for Crafting a Strong Introduction
- Be Engaging: Your opening should grab attention and encourage the reader to keep going.
- Be Clear: Avoid ambiguity—clearly state your research question and purpose.
- Provide Context: Background information is essential to help the reader understand the topic, but avoid overwhelming them with too much detail at this stage.
- Stay Focused: Keep the introduction concise but informative, setting the tone for the rest of your paper.
Literature Review
The literature review is where you showcase the existing research that relates to your topic. It's your chance to demonstrate your understanding of the academic conversation and position your research within that context.
Summarizing Existing Research
- Review relevant studies, theories, and findings that directly relate to your research question. This provides a foundation for your paper and shows that your study is grounded in the existing body of work.
Highlighting Key Studies
- Identify the most influential or significant research in your field. These are the works that have shaped the current understanding of your topic, and they should be emphasized in your review.
Identifying Gaps or Controversies
- Point out areas where there is limited research, conflicting findings, or ongoing debates. These gaps or discrepancies provide justification for your own research.
Establishing Your Research’s Relevance
- Explain how your research contributes to the field. Whether you’re addressing a gap, building on existing studies, or proposing something new, clearly indicate how your work fits into the larger picture.
Tips for a Strong Literature Review
- Stay Focused: Only include studies that are directly relevant to your research question. Avoid summarizing every piece of literature you've read.
- Be Critical: Don’t just summarize—critically assess the strengths and weaknesses of the studies you include.
- Organize Effectively: Structure your review in a logical order, grouping studies by themes, methodologies, or findings.
- Show Connections: Discuss how different studies relate to one another and to your research. This helps build a coherent narrative.
The methodology section details how you conducted your research. This is where you explain your approach, so others can understand and potentially replicate your study.
Research Design
- Outline the overall design of your study. Are you using qualitative, quantitative, or mixed methods? Define the type of research you're conducting (e.g., case study, survey, experiment).
Data Collection
- Explain how you gathered your data. Were interviews conducted? Surveys distributed? Or perhaps you collected data through observation or archival research. Be specific about the tools, instruments, or platforms you used.
Participants and Sampling
- If applicable, describe your sample group. Who participated in your study? How were they selected? Include details like the size of your sample and any inclusion/exclusion criteria.
Data Analysis
- Discuss how you analyzed your data. Did you use statistical methods, thematic analysis, coding, or another technique? Make sure to explain why these methods were appropriate for your research question.
Ethical Considerations
- Briefly mention any ethical protocols you followed, such as obtaining consent from participants or ensuring anonymity. If your research involved sensitive topics, this is especially important to address.
Tips for Writing Your Methodology
- Be Detailed but Clear: Provide enough detail so your methods can be understood or replicated, but avoid overloading with unnecessary jargon.
- Justify Your Choices: Explain why you chose specific methods over others and how they align with your research objectives.
- Stay Organized: Break your methodology into clear sections to improve readability and flow.
Results Tab
In the results section, you present the findings of your research. This is where you report what you discovered, without interpretation (that comes in the Discussion section). Clarity is key, especially if you are using visuals to support your findings.
Presentation of Data
- Clearly present your research results. This can include numerical data, text analysis, or findings from experiments, surveys, or interviews.
Use of Visuals
- Incorporate charts, tables, graphs, or other visuals to illustrate key points. Ensure that these visuals are well-labeled and easy to understand. Each visual should have a caption explaining what it represents.
Organizing Results
- Structure your results logically. You might choose to organize them by research question, themes, or hypotheses. Make sure there’s a clear flow, so readers can follow your findings easily.
Statistical or Analytical Reporting (if applicable)
- If you conducted statistical analysis, report your findings using appropriate measures (e.g., averages, standard deviations, significance levels). Be transparent about any statistical software or formulas used.
Relevant Findings Only
- Only include results that directly relate to your research question or hypothesis. Avoid tangents or irrelevant data.
Tips for a Clear Results Section
- Be Objective: This is not the place for interpretation—just present the facts.
- Visual Clarity: Ensure any visuals are clear, well-labeled, and directly support your results.
- Use Subheadings: If you have multiple results or sections, use subheadings to organize them.
- Stick to the Findings: Avoid analysis or speculation here; save that for the Discussion.
Discussion Tab
The discussion is where you interpret your findings. This is your opportunity to explain what the results mean, how they relate to your research question, and what implications they have for the field.
Interpretation of Results
- Explain what your results mean in the context of your research question. How do they answer the question or support (or refute) your hypothesis? Dive into the significance of the findings.
Connection to Existing Research
- Relate your findings back to the literature you reviewed earlier. How do your results compare with previous studies? Do they support or challenge existing theories?
Implications of the Study
- Discuss the broader implications of your research. What does it contribute to the field? Does it suggest changes in practice, policy, or further research avenues?
Limitations
- Acknowledge any limitations of your study. Were there constraints related to time, sample size, or methodology? Transparency about limitations adds credibility to your research.
Recommendations for Future Research
- Suggest areas where future researchers can explore. Perhaps there were aspects of the topic you couldn’t address fully, or new questions arose based on your findings.
Tips for a Strong Discussion Section
- Be Analytical: Focus on interpretation, not just re-stating results.
- Relate to Literature: Show how your findings fit within the broader research context.
- Be Honest About Limitations: Acknowledging weaknesses shows thoroughness and integrity.
- Highlight the Importance: Emphasize the practical or theoretical value of your work.
The conclusion ties everything together. It should succinctly summarize your key findings, emphasize their significance, and leave the reader with a clear understanding of what you’ve contributed to the field.
Summary of Key Findings
- Briefly restate your most important results. Focus on the findings that directly answer your research question and highlight their relevance.
Restating the Research Question/Thesis
- Revisit your original research question or thesis and clearly explain how your findings address it.
Implications and Impact
- Reinforce the broader significance of your work. How do your findings contribute to the academic field or practical applications? This is your chance to leave a lasting impression.
Recommendations for Future Research or Practice
- Suggest directions for future studies or practical steps that can be taken based on your findings. This ensures your conclusion looks forward rather than simply wrapping up.
Final Thought/Call to Action
- End on a strong note! Offer a thought-provoking statement, reflection, or call to action, encouraging further discussion or research.
Tips for a Strong Conclusion
- Be Concise: Keep it focused—summarize, don’t rehash.
- Be Forward-Looking: Emphasize the impact and potential future directions.
- End with Confidence: Leave readers with a clear understanding of your research's importance.
How to Create a Clearly Structured Paper
Provides a step-by-step guide to organizing an effective essay or research paper outline, focusing on creating clear, logical sections that streamline the writing process.
- << Previous: Literature Reviews
- Next: Citations >>
- Last Updated: Sep 20, 2024 3:27 PM
- URL: https://tsu.libguides.com/researchmethods
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Research paper
Writing a Research Paper Introduction | Step-by-Step Guide
Published on September 24, 2022 by Jack Caulfield . Revised on September 5, 2024.

The introduction to a research paper is where you set up your topic and approach for the reader. It has several key goals:
- Present your topic and get the reader interested
- Provide background or summarize existing research
- Position your own approach
- Detail your specific research problem and problem statement
- Give an overview of the paper’s structure
The introduction looks slightly different depending on whether your paper presents the results of original empirical research or constructs an argument by engaging with a variety of sources.
The five steps in this article will help you put together an effective introduction for either type of research paper.
Instantly correct all language mistakes in your text
Upload your document to correct all your mistakes in minutes
Table of contents
Step 1: introduce your topic, step 2: describe the background, step 3: establish your research problem, step 4: specify your objective(s), step 5: map out your paper, research paper introduction examples, frequently asked questions about the research paper introduction.
The first job of the introduction is to tell the reader what your topic is and why it’s interesting or important. This is generally accomplished with a strong opening hook.
The hook is a striking opening sentence that clearly conveys the relevance of your topic. Think of an interesting fact or statistic, a strong statement, a question, or a brief anecdote that will get the reader wondering about your topic.
For example, the following could be an effective hook for an argumentative paper about the environmental impact of cattle farming:
A more empirical paper investigating the relationship of Instagram use with body image issues in adolescent girls might use the following hook:
Don’t feel that your hook necessarily has to be deeply impressive or creative. Clarity and relevance are still more important than catchiness. The key thing is to guide the reader into your topic and situate your ideas.
Prevent plagiarism. Run a free check.
This part of the introduction differs depending on what approach your paper is taking.
In a more argumentative paper, you’ll explore some general background here. In a more empirical paper, this is the place to review previous research and establish how yours fits in.
Argumentative paper: Background information
After you’ve caught your reader’s attention, specify a bit more, providing context and narrowing down your topic.
Provide only the most relevant background information. The introduction isn’t the place to get too in-depth; if more background is essential to your paper, it can appear in the body .
Empirical paper: Describing previous research
For a paper describing original research, you’ll instead provide an overview of the most relevant research that has already been conducted. This is a sort of miniature literature review —a sketch of the current state of research into your topic, boiled down to a few sentences.
This should be informed by genuine engagement with the literature. Your search can be less extensive than in a full literature review, but a clear sense of the relevant research is crucial to inform your own work.
Begin by establishing the kinds of research that have been done, and end with limitations or gaps in the research that you intend to respond to.
The next step is to clarify how your own research fits in and what problem it addresses.
Argumentative paper: Emphasize importance
In an argumentative research paper, you can simply state the problem you intend to discuss, and what is original or important about your argument.
Empirical paper: Relate to the literature
In an empirical research paper, try to lead into the problem on the basis of your discussion of the literature. Think in terms of these questions:
- What research gap is your work intended to fill?
- What limitations in previous work does it address?
- What contribution to knowledge does it make?
You can make the connection between your problem and the existing research using phrases like the following.
| Although has been studied in detail, insufficient attention has been paid to . | You will address a previously overlooked aspect of your topic. |
| The implications of study deserve to be explored further. | You will build on something suggested by a previous study, exploring it in greater depth. |
| It is generally assumed that . However, this paper suggests that … | You will depart from the consensus on your topic, establishing a new position. |
Now you’ll get into the specifics of what you intend to find out or express in your research paper.
The way you frame your research objectives varies. An argumentative paper presents a thesis statement, while an empirical paper generally poses a research question (sometimes with a hypothesis as to the answer).
Argumentative paper: Thesis statement
The thesis statement expresses the position that the rest of the paper will present evidence and arguments for. It can be presented in one or two sentences, and should state your position clearly and directly, without providing specific arguments for it at this point.
Empirical paper: Research question and hypothesis
The research question is the question you want to answer in an empirical research paper.
Present your research question clearly and directly, with a minimum of discussion at this point. The rest of the paper will be taken up with discussing and investigating this question; here you just need to express it.
A research question can be framed either directly or indirectly.
- This study set out to answer the following question: What effects does daily use of Instagram have on the prevalence of body image issues among adolescent girls?
- We investigated the effects of daily Instagram use on the prevalence of body image issues among adolescent girls.
If your research involved testing hypotheses , these should be stated along with your research question. They are usually presented in the past tense, since the hypothesis will already have been tested by the time you are writing up your paper.
For example, the following hypothesis might respond to the research question above:
Don't submit your assignments before you do this
The academic proofreading tool has been trained on 1000s of academic texts. Making it the most accurate and reliable proofreading tool for students. Free citation check included.
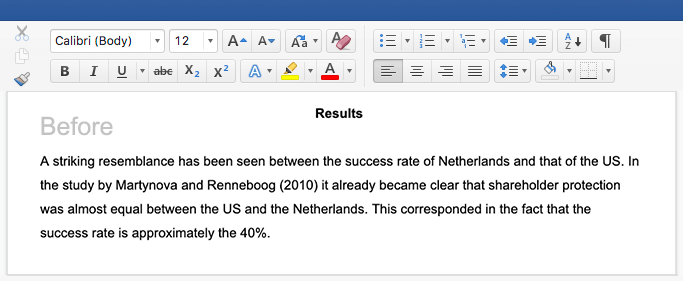
Try for free
The final part of the introduction is often dedicated to a brief overview of the rest of the paper.
In a paper structured using the standard scientific “introduction, methods, results, discussion” format, this isn’t always necessary. But if your paper is structured in a less predictable way, it’s important to describe the shape of it for the reader.
If included, the overview should be concise, direct, and written in the present tense.
- This paper will first discuss several examples of survey-based research into adolescent social media use, then will go on to …
- This paper first discusses several examples of survey-based research into adolescent social media use, then goes on to …
Scribbr’s paraphrasing tool can help you rephrase sentences to give a clear overview of your arguments.
Full examples of research paper introductions are shown in the tabs below: one for an argumentative paper, the other for an empirical paper.
- Argumentative paper
- Empirical paper
Are cows responsible for climate change? A recent study (RIVM, 2019) shows that cattle farmers account for two thirds of agricultural nitrogen emissions in the Netherlands. These emissions result from nitrogen in manure, which can degrade into ammonia and enter the atmosphere. The study’s calculations show that agriculture is the main source of nitrogen pollution, accounting for 46% of the country’s total emissions. By comparison, road traffic and households are responsible for 6.1% each, the industrial sector for 1%. While efforts are being made to mitigate these emissions, policymakers are reluctant to reckon with the scale of the problem. The approach presented here is a radical one, but commensurate with the issue. This paper argues that the Dutch government must stimulate and subsidize livestock farmers, especially cattle farmers, to transition to sustainable vegetable farming. It first establishes the inadequacy of current mitigation measures, then discusses the various advantages of the results proposed, and finally addresses potential objections to the plan on economic grounds.
The rise of social media has been accompanied by a sharp increase in the prevalence of body image issues among women and girls. This correlation has received significant academic attention: Various empirical studies have been conducted into Facebook usage among adolescent girls (Tiggermann & Slater, 2013; Meier & Gray, 2014). These studies have consistently found that the visual and interactive aspects of the platform have the greatest influence on body image issues. Despite this, highly visual social media (HVSM) such as Instagram have yet to be robustly researched. This paper sets out to address this research gap. We investigated the effects of daily Instagram use on the prevalence of body image issues among adolescent girls. It was hypothesized that daily Instagram use would be associated with an increase in body image concerns and a decrease in self-esteem ratings.
The introduction of a research paper includes several key elements:
- A hook to catch the reader’s interest
- Relevant background on the topic
- Details of your research problem
and your problem statement
- A thesis statement or research question
- Sometimes an overview of the paper
Don’t feel that you have to write the introduction first. The introduction is often one of the last parts of the research paper you’ll write, along with the conclusion.
This is because it can be easier to introduce your paper once you’ve already written the body ; you may not have the clearest idea of your arguments until you’ve written them, and things can change during the writing process .
The way you present your research problem in your introduction varies depending on the nature of your research paper . A research paper that presents a sustained argument will usually encapsulate this argument in a thesis statement .
A research paper designed to present the results of empirical research tends to present a research question that it seeks to answer. It may also include a hypothesis —a prediction that will be confirmed or disproved by your research.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
Caulfield, J. (2024, September 05). Writing a Research Paper Introduction | Step-by-Step Guide. Scribbr. Retrieved September 22, 2024, from https://www.scribbr.com/research-paper/research-paper-introduction/
Is this article helpful?

Jack Caulfield
Other students also liked, writing strong research questions | criteria & examples, writing a research paper conclusion | step-by-step guide, research paper format | apa, mla, & chicago templates, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts
How to Write a Research Paper

If you already have a headache trying to understand what research paper is all about, we have created an ultimate guide for you on how to write a research paper. You will find all the answers to your questions regarding structure, planning, doing investigation, finding the topic that appeals to you. Plus, you will find out the secret to an excellent paper. Are you at the edge of your seat? Let us start with the basics then.
- What is a Research Paper
- Reasons for Writing a Research Paper
- Report Papers and Thesis Papers
- How to Start a Research Paper
- How to Choose a Topic for a Research Paper
- How to Write a Proposal for a Research Paper
- How to Write a Research Plan
- How to Do Research
- How to Write an Outline for a Research Paper
- How to Write a Thesis Statement for a Research Paper
- How to Write a Research Paper Rough Draft
- How to Write an Introduction for a Research Paper
- How to Write a Body of a Research Paper
- How to Write a Conclusion for a Research Paper
- How to Write an Abstract for a Research Paper
- How to Revise and Edit a Research Paper
- How to Write a Bibliography for a Research Paper
- What Makes a Good Research Paper
Research Paper Writing Services
What is a research paper.

Academic Writing, Editing, Proofreading, And Problem Solving Services
Get 10% off with 24start discount code.
You probably know the saying ‘the devil is not as black as he is painted’. This particular saying is absolutely true when it comes to writing a research paper. Your feet are cold even with the thought of this assignment. You have heard terrifying stories from older students. You have never done this before, so certainly you are scared. What is a research paper? How should I start? What are all these requirements about?
Luckily, you have a friend in need. That is our writing service. First and foremost, let us clarify the definition. A research paper is a piece of academic writing that provides information about a particular topic that you’ve researched . In other words, you choose a topic: about historical events, the work of some artist, some social issues etc. Then you collect data on the given topic and analyze it. Finally, you put your analysis on paper. See, it is not as scary as it seems. If you are still having doubts, whether you can handle it yourself, we are here to help you. Our team of writers can help you choose the topic, or give you advice on how to plan your work, or how to start, or craft a paper for you. Just contact us 24/7 and see everything yourself.
5 Reasons for Writing a Research Paper
Why should I spend my time writing some academic paper? What is the use of it? Is not some practical knowledge more important? The list of questions is endless when it comes to a research paper. That is why we have outlined 5 main reasons why writing a research paper is a good thing.
- You will learn how to organize your time
If you want to write a research paper, you will have to learn how to manage your time. This type of assignment cannot be done overnight. It requires careful planning and you will need to learn how to do it. Later, you will be able to use these time-managing skills in your personal life, so why not developing them?
- You will discover your writing skills
You cannot know something before you try it. This rule relates to writing as well. You cannot claim that you cannot write until you try it yourself. It will be really difficult at the beginning, but then the words will come to your head themselves.
- You will improve your analytical skills
Writing a research paper is all about investigation and analysis. You will need to collect data, examine and classify it. These skills are needed in modern life more than anything else is.
- You will gain confidence
Once you do your own research, it gives you the feeling of confidence in yourself. The reason is simple human brain likes solving puzzles and your assignment is just another puzzle to be solved.
- You will learn how to persuade the reader
When you write your paper, you should always remember that you are writing it for someone to read. Moreover, you want this someone to believe in your ideas. For this reason, you will have to learn different convincing methods and techniques. You will learn how to make your writing persuasive. In turns, you will be able to use these methods in real life.
What is the Difference between Report and Thesis Papers?
A common question is ‘what is the difference between a report paper and a thesis paper?’ The difference lies in the aim of these two assignments. While the former aims at presenting the information, the latter aims at providing your opinion on the matter. In other words, in a report paper you have to summarize your findings. In a thesis paper, you choose some issue and defend your point of view by persuading the reader. It is that simple.
A thesis paper is a more common assignment than a report paper. This task will help a professor to evaluate your analytical skills and skills to present your ideas logically. These skills are more important than just the ability to collect and summarize data.
How to Write a Research Paper Step by Step
Research comes from the French word rechercher , meaning “to seek out.” Writing a research paper requires you to seek out information about a subject, take a stand on it, and back it up with the opinions, ideas, and views of others. What results is a printed paper variously known as a term paper or library paper, usually between five and fifteen pages long—most instructors specify a minimum length—in which you present your views and findings on the chosen subject.

It is not a secret that the majority of students hate writing a research paper. The reason is simple it steals your time and energy. Not to mention, constant anxiety that you will not be able to meet the deadline or that you will forget about some academic requirement.
We will not lie to you; a research paper is a difficult assignment. You will have to spend a lot of time. You will need to read, to analyze, and to search for the material. You will probably be stuck sometimes. However, if you organize your work smart, you will gain something that is worth all the effort – knowledge, experience, and high grades.
The reason why many students fail writing a research paper is that nobody explained them how to start and how to plan their work. Luckily, you have found our writing service and we are ready to shed the light on this dark matter.
We have created a step by step guide for you on how to write a research paper. We will dwell upon the structure, the writing tips, the writing strategies as well as academic requirements. Read this whole article and you will see that you can handle writing this assignment and our team of writers is here to assist you.
How to Start a Research Paper?

It all starts with the assignment. Your professor gives you the task. It may be either some general issue or specific topic to write about. Your assignment is your first guide to success. If you understand what you need to do according to the assignment, you are on the road to high results. Do not be scared to clarify your task if you need to. There is nothing wrong in asking a question if you want to do something right. You can ask your professor or you can ask our writers who know a thing or two in academic writing.
It is essential to understand the assignment. A good beginning makes a good ending, so start smart.
Learn how to start a research paper .
Choosing a Topic for a Research Paper

We have already mentioned that it is not enough to do great research. You need to persuade the reader that you have made some great research. What convinces better that an eye-catching topic? That is why it is important to understand how to choose a topic for a research paper.
First, you need to delimit the general idea to a more specific one. Secondly, you need to find what makes this topic interesting for you and for the academia. Finally, you need to refine you topic. Remember, it is not something you will do in one day. You can be reshaping your topic throughout your whole writing process. Still, reshaping not changing it completely. That is why keep in your head one main idea: your topic should be precise and compelling .
Learn how to choose a topic for a research paper .
How to Write a Proposal for a Research Paper?

If you do not know what a proposal is, let us explain it to you. A proposal should answer three main questions:
- What is the main aim of your investigation?
- Why is your investigation important?
- How are you going to achieve the results?
In other words, proposal should show why your topic is interesting and how you are going to prove it. As to writing requirements, they may differ. That is why make sure you find out all the details at your department. You can ask your departmental administrator or find information online at department’s site. It is crucial to follow all the administrative requirements, as it will influence your grade.
Learn how to write a proposal for a research paper .
How to Write a Research Plan?

The next step is writing a plan. You have already decided on the main issues, you have chosen the bibliography, and you have clarified the methods. Here comes the planning. If you want to avoid writer’s block, you have to structure you work. Discuss your strategies and ideas with your instructor. Think thoroughly why you need to present some data and ideas first and others second. Remember that there are basic structure elements that your research paper should include:
- Thesis Statement
- Introduction
- Bibliography
You should keep in mind this skeleton when planning your work. This will keep your mind sharp and your ideas will flow logically.
Learn how to write a research plan .
How to Do Research?

Your research will include three stages: collecting data, reading and analyzing it, and writing itself.
First, you need to collect all the material that you will need for you investigation: films, documents, surveys, interviews, and others. Secondly, you will have to read and analyze. This step is tricky, as you need to do this part smart. It is not enough just to read, as you cannot keep in mind all the information. It is essential that you make notes and write down your ideas while analyzing some data. When you get down to the stage number three, writing itself, you will already have the main ideas written on your notes. Plus, remember to jot down the reference details. You will then appreciate this trick when you will have to write the bibliography.
If you do your research this way, it will be much easier for you to write the paper. You will already have blocks of your ideas written down and you will just need to add some material and refine your paper.
Learn how to do research .
How to Write an Outline for a Research Paper?

To make your paper well organized you need to write an outline. Your outline will serve as your guiding star through the writing process. With a great outline you will not get sidetracked, because you will have a structured plan to follow. Both you and the reader will benefit from your outline. You present your ideas logically and you make your writing coherent according to your plan. As a result, this outline guides the reader through your paper and the reader enjoys the way you demonstrate your ideas.
Learn how to write an outline for a research paper . See research paper outline examples .
How to Write a Thesis Statement for a Research Paper?

Briefly, the thesis is the main argument of your research paper. It should be precise, convincing and logical. Your thesis statement should include your point of view supported by evidence or logic. Still, remember it should be precise. You should not beat around the bush, or provide all the possible evidence you have found. It is usually a single sentence that shows your argument. In on sentence you should make a claim, explain why it significant and convince the reader that your point of view is important.
Learn how to write a thesis statement for a research paper . See research paper thesis statement examples .
Should I Write a Rough Draft for a Research Paper?

Do you know any writer who put their ideas on paper, then never edited them and just published? Probably, no writer did so. Writing a research paper is no exception. It is impossible to cope with this assignment without writing a rough draft.
Your draft will help you understand what you need to polish to make your paper perfect. All the requirements, academic standards make it difficult to do everything flawlessly at the first attempt. Make sure you know all the formatting requirements: margins, words quantity, reference requirements, formatting styles etc.
Learn how to write a rough draft for a research paper .
How to Write an Introduction for a Research Paper?

Let us make it more vivid for you. We have narrowed down the tips on writing an introduction to the three main ones:
- Include your thesis in your introduction
Remember to include the thesis statement in your introduction. Usually, it goes at the end of the first paragraph.
- Present the main ideas of the body
You should tell the main topics you are going to discuss in the main body. For this reason, before writing this part of introduction, make sure you know what is your main body is going to be about. It should include your main ideas.
- Polish your thesis and introduction
When you finish the main body of your paper, come back to the thesis statement and introduction. Restate something if needed. Just make it perfect; because introduction is like the trailer to your paper, it should make the reader want to read the whole piece.
Learn how to write an introduction for a research paper . See research paper introduction examples .
How to Write a Body of a Research Paper?

A body is the main part of your research paper. In this part, you will include all the needed evidence; you will provide the examples and support your argument.
It is important to structure your paragraphs thoroughly. That is to say, topic sentence and the evidence supporting the topic. Stay focused and do not be sidetracked. You have your outline, so follow it.
Here are the main tips to keep in head when writing a body of a research paper:
- Let the ideas flow logically
- Include only relevant information
- Provide the evidence
- Structure the paragraphs
- Make the coherent transition from one paragraph to another
See? When it is all structured, it is not as scary as it seemed at the beginning. Still, if you have doubts, you can always ask our writers for help.
Learn how to write a body of a research paper . See research paper transition examples .
How to Write a Conclusion for a Research Paper?

Writing a good conclusion is important as writing any other part of the paper. Remember that conclusion is not a summary of what you have mentioned before. A good conclusion should include your last strong statement.
If you have written everything according to the plan, the reader already knows why your investigation is important. The reader has already seen the evidence. The only thing left is a strong concluding thought that will organize all your findings.
Never include any new information in conclusion. You need to conclude, not to start a new discussion.
Learn how to write a conclusion for a research paper .
How to Write an Abstract for a Research Paper?

An abstract is a brief summary of your paper, usually 100-200 words. You should provide the main gist of your paper in this short summary. An abstract can be informative, descriptive or proposal. Depending on the type of abstract, you need to write, the requirements will differ.
To write an informative abstract you have to provide the summary of the whole paper. Informative summary. In other words, you need to tell about the main points of your work, the methods used, the results and the conclusion of your research.
To write a descriptive abstract you will not have to provide any summery. You should write a short teaser of your paper. That is to say, you need to write an overview of your paper. The aim of a descriptive abstract is to interest the reader.
Finally, to write a proposal abstract you will need to write the basic summary as for the informative abstract. However, the difference is the following: you aim at persuading someone to let you write on the topic. That is why, a proposal abstract should present your topic as the one worth investigating.
Learn how to write an abstract for a research paper .
Should I Revise and Edit a Research Paper?

Revising and editing your paper is essential if you want to get high grades. Let us help you revise your paper smart:
- Check your paper for spelling and grammar mistakes
- Sharpen the vocabulary
- Make sure there are no slang words in your paper
- Examine your paper in terms of structure
- Compare your topic, thesis statement to the whole piece
- Check your paper for plagiarism
If you need assistance with proofreading and editing your paper, you can turn to the professional editors at our service. They will help you polish your paper to perfection.
Learn how to revise and edit a research paper .
How to Write a Bibliography for a Research Paper?

First, let us make it clear that bibliography and works cited are two different things. Works cited are those that you cited in your paper. Bibliography should include all the materials you used to do your research. Still, remember that bibliography requirements differ depending on the formatting style of your paper. For this reason, make sure you ask you professor all the requirements you need to meet to avoid any misunderstanding.
Learn how to write a bibliography for a research paper .
The Key Secret to a Good Research Paper
Now when you know all the stages of writing a research paper, you are ready to find the key to a good research paper:
- Choose the topic that really interests you
- Make the topic interesting for you even if it is not at the beginning
- Follow the step by step guide and do not get sidetracked
- Be persistent and believe in yourself
- Really do research and write your paper from scratch
- Learn the convincing writing techniques and use them
- Follow the requirements of your assignment
- Ask for help if needed from real professionals
Feeling more confident about your paper now? We are sure you do. Still, if you need help, you can always rely on us 24/7.
We hope we have made writing a research paper much easier for you. We realize that it requires lots of time and energy. We believe when you say that you cannot handle it anymore. For this reason, we have been helping students like you for years. Our professional team of writers is ready to tackle any challenge.
All our authors are experienced writers crafting excellent academic papers. We help students meet the deadline and get the top grades they want. You can see everything yourself. All you need to do is to place your order online and we will contact you. Writing a research paper with us is truly easy, so why do not you check it yourself?
Additional Resources for Research Paper Writing:
- Anthropology Research
- Career Research
- Communication Research
- Criminal Justice Research
- Health Research
- Political Science Research
- Psychology Research
- Sociology Research
ORDER HIGH QUALITY CUSTOM PAPER

Writing a Research Paper
This page lists some of the stages involved in writing a library-based research paper.
Although this list suggests that there is a simple, linear process to writing such a paper, the actual process of writing a research paper is often a messy and recursive one, so please use this outline as a flexible guide.
Discovering, Narrowing, and Focusing a Researchable Topic
- Try to find a topic that truly interests you
- Try writing your way to a topic
- Talk with your course instructor and classmates about your topic
- Pose your topic as a question to be answered or a problem to be solved
Finding, Selecting, and Reading Sources
You will need to look at the following types of sources:
- library catalog, periodical indexes, bibliographies, suggestions from your instructor
- primary vs. secondary sources
- journals, books, other documents
Grouping, Sequencing, and Documenting Information
The following systems will help keep you organized:
- a system for noting sources on bibliography cards
- a system for organizing material according to its relative importance
- a system for taking notes
Writing an Outline and a Prospectus for Yourself
Consider the following questions:
- What is the topic?
- Why is it significant?
- What background material is relevant?
- What is my thesis or purpose statement?
- What organizational plan will best support my purpose?
Writing the Introduction
In the introduction you will need to do the following things:
- present relevant background or contextual material
- define terms or concepts when necessary
- explain the focus of the paper and your specific purpose
- reveal your plan of organization
Writing the Body
- Use your outline and prospectus as flexible guides
- Build your essay around points you want to make (i.e., don’t let your sources organize your paper)
- Integrate your sources into your discussion
- Summarize, analyze, explain, and evaluate published work rather than merely reporting it
- Move up and down the “ladder of abstraction” from generalization to varying levels of detail back to generalization
Writing the Conclusion
- If the argument or point of your paper is complex, you may need to summarize the argument for your reader.
- If prior to your conclusion you have not yet explained the significance of your findings or if you are proceeding inductively, use the end of your paper to add your points up, to explain their significance.
- Move from a detailed to a general level of consideration that returns the topic to the context provided by the introduction.
- Perhaps suggest what about this topic needs further research.
Revising the Final Draft
- Check overall organization : logical flow of introduction, coherence and depth of discussion in body, effectiveness of conclusion.
- Paragraph level concerns : topic sentences, sequence of ideas within paragraphs, use of details to support generalizations, summary sentences where necessary, use of transitions within and between paragraphs.
- Sentence level concerns: sentence structure, word choices, punctuation, spelling.
- Documentation: consistent use of one system, citation of all material not considered common knowledge, appropriate use of endnotes or footnotes, accuracy of list of works cited.

Academic and Professional Writing
This is an accordion element with a series of buttons that open and close related content panels.
Analysis Papers
Reading Poetry
A Short Guide to Close Reading for Literary Analysis
Using Literary Quotations
Play Reviews
Writing a Rhetorical Précis to Analyze Nonfiction Texts
Incorporating Interview Data
Grant Proposals
Planning and Writing a Grant Proposal: The Basics
Additional Resources for Grants and Proposal Writing
Job Materials and Application Essays
Writing Personal Statements for Ph.D. Programs
- Before you begin: useful tips for writing your essay
- Guided brainstorming exercises
- Get more help with your essay
- Frequently Asked Questions
Resume Writing Tips
CV Writing Tips
Cover Letters
Business Letters
Proposals and Dissertations
Resources for Proposal Writers
Resources for Dissertators
Research Papers
Planning and Writing Research Papers
Quoting and Paraphrasing
Writing Annotated Bibliographies
Creating Poster Presentations
Writing an Abstract for Your Research Paper
Thank-You Notes
Advice for Students Writing Thank-You Notes to Donors
Reading for a Review
Critical Reviews
Writing a Review of Literature
Scientific Reports
Scientific Report Format
Sample Lab Assignment
Writing for the Web
Writing an Effective Blog Post
Writing for Social Media: A Guide for Academics

- Gradehacker
- Meet the Team
- Fast-Track Degree Support
- Class Assistance
- Learning Center
- Gradehacker TV
- Fill a Request
- Book a Call
- Video Tutorials
- YouTube Video Tutorials
How to Write a Research Paper | The Ultimate Step-by-Step Guide
Santiago mallea.
- Writing Tips

Content Writer At Gradehacker
- March, 2024
What Are the Steps for Writing a Research Paper?
There’s not a specific way to write academic papers. However, we will share the nine steps that will guarantee a great result:
1. Choose Your Research Paper Topic
2. do your research, 3. write your thesis statement, 4. create your paper outline, 5. write the introduction, 6. write your body, 7. write your conclusion, 8. add your paper format style, 9. revise and edit your paper.
As one of the most common and important assignments you’ll find throughout your college experience, knowing how to write a research paper is highly necessary.
Coming up with a good topic can be hard sometimes; you could look for a variety of sources and gather interesting information, but it still can seem to be a tedious and complex process to master if you don’t have the right understanding of the process.
Luckily, here at Gradehacker, we have years of experience helping our clients as the non-traditional student #1 resource
That’s why we are sharing a clear and easy-to-follow guide on how to write a research paper with our pro tips that will help you make a difference when writing.

What Is a Research Paper?
A research paper is a type of academic writing that involves information analysis, source evaluation, and argument explanation on a specific topic.
They are not the same as academic essays and argumentative papers since they are usually longer, more detailed, and rely more on statistics and pre-existing research than on strong opinions.
This assignment aims to prove both your writing and researching skills by demonstrating a clear understanding of the topic, engaging with all types of sources, and making an original contribution to the field of study.
While research papers are common among many branches of the academic community as a way to share information, they are also one of the most typical assignments required in colleges.
Regardless of which career you are following, whether it’s nursing, economics, business, and even social sciences, there will come a time when you’ll have to write a research paper.
But don’t worry, here is our step-by-step guide:
As you’ll see, the first three steps are slightly related and are key to the entire process. But the first thing you have to do is choose a good and interesting research paper topic.
Always check first what the instructions of your assignments say. See if there are any specifications regarding which topic you have to choose or if there are any limitations to it.
Once you know for sure which is the field of study you’ll be conducting the research on, think of a topic that is:
- Interesting to you
- Possible to research
It must be ideally of interest because writing a research paper takes time, and spending so many hours on something you find boring will make the entire process tedious, making everything worse.
By picking something you like, you’ll have a more pleasant time and be more naturally curious about it. The more interested you are, the more information you look for, and the more you understand the topic!
If you have trouble finding an original topic, try starting with a broad subject area and then narrowing it down to its subtopics. You’ll find more unique subjects, which will be more specific to write about.
Lastly, it must be a topic that is possible to research. For instance, if you want to write about how the population of sea turtles varied between the 1800s and 1900s, but you don’t have access to factual data and statistics to conduct your research, then you should look for a possible topic.
However, as this first step relies so heavily on what information you have found, it goes hand in hand with the second step:
Now, one of the most important, if not the most important, parts of the process is conducting the research.
Once you know which topic you’ll be writing about, you need to search as many sources as possible. You may spend at least two hours skimming through many scholarly articles that could add to your paper.
It can take time, but it will be worth it once you start the writing process. You’ll see why in the fourth step.
Which is the best way to conduct the research?
Easy: read as many academic sources as you can. Here are a few tips to have in mind to improve your research experience:
Read the abstract
Many scholarly sources have an abstract. They summarize the paper’s content, with the main statistics, observations, and their conclusion.
Save time and read the abstract first; if you found at least one helpful sentence, keep reading and find out more.
Save each source
No matter how long the information is or whether it supports or opposes your main argument, save everything that is at least related to your topic.
Save statistics, facts, results from surveys, and studies’ conclusions, even those you think you won’t need to use.
It can come in handy if, during writing, you suddenly change your mind and realize that the statistic you randomly read in a source is now useful; then you’ll know where to find it.
Don't be afraid of using the sources that others are citing
While you are doing research, something that can happen is that you find an interesting piece of information only to see that the primary source you are reading is citing it from another place.
In case that primary source has other original data you can use but still need to use the other source, you go ahead and use both, each for its respective information!
Know where to look for
Finding peer-reviewed and trustworthy papers can be hard.
You can trust sites like NCBI and ResearchGate , which hold various free sources approved by the community. And don’t forget to check what you can find in your college’s library!
So, while you are doing your research, you’ll have enough information to start with step three:
By having all the gathered information, you can develop a specific and robust thesis statement. This is your central argument , your main claim that establishes the purpose and position of your essay.
You will have to include it in the introduction, but more on that in step five.
For now, it’s important that your thesis statement summarizes your argument in one or two sentences and includes all the main arguments of the discussion without getting too much into details. It has to be concise and coherent.
Coming up with a thesis statement is actually easy; you only need to answer the question that made you start the research.
For example, let’s say you are approaching mental health issues from a racial perspective and the question that kicked off your research was: “Were the health policies implemented in Latino communities to warn about the importance of mental health useful”?
If your research concluded that it mainly worked in regions where Latino families lived in America for more than ten years, then this would be a good basis for your thesis statement.

With all of the information gathered, you can start structuring your ideas into an outline.
Here is when the thinking process begins, as you’ll have to organize all the ideas and information in a cohesive order.
Usually, research paper assignments require you to include an outline. If it’s not specified or you are unsure about it, you should ask your professor. However, you should still create one, as it will help you structure all the topics you will cover in one place , serving as your guiding roadmap during the writing process.
First, you have to list all the important categories and their subtopics.
Then, rearrange these categories in an order that is connected and makes sense. You can ask yourself these questions as a guide:
- Which topics are related?
- Which topics need to be explained first in order to talk about another topic?
- Is there any topic that doesn't add to the cohesion if it's presented out of sequence?
- Which subtopics are the most important?
- Is any subtopic related to a subtopic of another main topic?
Once you have placed all your categories in a cohesive order, create your roadmap by adding sentences and even paragraphs to your outline. Add the purpose of each specific topic and subtopic, and include which sources you’ll be using in that paragraph.
By doing this, you are already starting to write your research paper.
If you want to know how to make the best research paper outline, check out our article and learn the best tips!
Now that you have all the sources gathered, the thesis statement done, and the outline ready, you can start writing the introduction.
This step shouldn’t be hard if you are paying attention and already have your thesis statement.
The purpose of this first paragraph is to bring context to the reader. Introduce them to the field of study, give them context on the issue you are writing, and tell them the thesis statement that will be the main point of discussion throughout the entire paper.
Don’t include details or cite sources; leave that for the body. The introduction must be short and clear. We recommend between 80 and 150 words, depending on the overall length of your assignment.
Use your thesis statement to open your paper and tell the reader which will be the specific and original topic you will be studying.

The body of your paper is where you’ll develop your thesis by explaining and analyzing all the information and statistics you got from your sources.
The easiest way to start writing is by following the roadmap you have from step four. You have already divided your body into sections by having your outline prepared. Now, you just need to start writing and linking everything in a natural and cohesive structure.
Remember, your outline is flexible, meaning that you can change the order or the overall structure while you are writing if you think this is for the best.
When writing, make sure that you only cover one idea per paragraph. Instead of gathering too much different information in one lengthy section, explain each topic or subtopic individually.
This way, you’ll have a better-structured paper that is easy to read and allows readers to find faster valuable information they might be looking for.
To maintain a good flow while clearly dividing the paper into sections, you need to use connectors whenever you start writing on a new topic.
To add information, use:
- Furthermore
- In addition to this
To change the topic and explain an argument that opposes the previous topic, use the following:
- Nevertheless
- Even though
- Nonetheless
Connectors are powerful and necessary elements of academic writing; you should always use them.
To finish your essay, you need a strong conclusion. You have to restate your thesis statement, list your main evidence, and briefly summarize what you have found.
Think of your conclusion as the only piece of information that the reader will remember from your paper.
Besides giving a sense of finality and restating the main points of your essay, you can also discuss the general consequences of your argument by suggesting that further research needs to be conducted on another specific topic.
Finally, don’t add any new information or details in the conclusion . It may seem obvious, but it’s actually one of the most common mistakes in academic writing.
Avoid this by knowing beforehand where you’ll be placing all the information you have gathered from the source, so you don’t get to the conclusion with valuable statistics that you forgot to include.
We haven’t forgotten about the formatting style. First, check on your assignment guidelines to see which format is required.
Depending on the one you have to use, you may have to include a Title Page at the beginning, but in every case, you’ll have to add a Reference List with all the sources you have used , following the proper citation style.
The most common formats are APA and MLA .
MLA uses in-text citations with the author’s last name and page number, while APA uses parenthetical citations with the author’s last name, year, and page number.
Then the header in MLA includes the author’s last name and page number, while in APA it includes a shortened title and page number. MLA typically doesn’t require a separate title page, while APA does.
Both formats use hanging indentation for the reference list entries.
We are not going to lie. The final step of the process may be a bit tedious, but it’s indispensable.
Once you have finished writing, you have to revise your paper for grammar, spelling, and stylistic mistakes and edit them when you find these errors. Yes, you will certainly find a few of them, at least.
It’s impossible to finish with a perfect first draft, regardless of how much experience you have written. So, you must read what you wrote from the start and see in detail what can be improved. Of course, check for grammar mistakes, but also be attentive to word repetitions and wordy sentences and see what changes you can make to your paper to enhance the overall result.
Don’t be closed to the idea of rewriting an entire paragraph or changing the order of your body sections.
Here are a few questions you can ask yourself while writing:
- Is the thesis statement clear?
- Is the structure well-organized and flows cohesively?
- Is the information appropriately cited?
- Does the information help support my thesis statement?
- Is the information I am using detailed and specific?
- Am I following the correct rules of the formatting style I have to use?
- Am I plagiarizing?
- Are the grammar, spelling, and punctuation correct?
The best way to check this is by reading your paper aloud or having somebody else read it while you hear it. When you hear, you’ll see that you’ll find technical mistakes and think of some improvements you can make.

Tips: Mastering Your Research Paper
Do and organize all the research before you start writing.
If you have all the information you need from the start, it will be easier to structure your paper. You should organize each source into categories.
Know which are your main sources with essential information , but also save all the secondary sources that may have less significant information that could add support to one specific argument. The more sources you save, the better.
Even if you don’t use all of them or later need to search for more, having a good amount of primary and secondary sources from the start will help you structure your essay better and faster.
Add an estimated length for each paragraph
While the length of your assignment will depend on every case, you should have an approximation of how many words you should dedicate to each paragraph to have a better idea of how much time you should spend with each.
It doesn’t matter if you later end up writing more on one and less on another; the number you set doesn’t have to be mandatory; instead, it serves as an estimated length for you to have in mind to track your progress.
You can save the introduction for last!
While it can be easy to write the introduction if you already have the thesis statement, sometimes you can spend too much time looking at a blank page because you are unsure how to start. So, you can leave it for last.
Start writing the body of your text , and by the time you get to the conclusion, you’ll have a better idea of which topics you have covered and what the main focus of your paper is, making it easier for you to finally write the introduction.

Use Grammarly
To check grammar mistakes, see stylistic suggestions, avoid repeated words, and have a percentage of plagiarized content , we recommend you use Grammarly. It’s a freemium app that works as your own online editor.
If you want more information on how Grammarly works, you can read our honest review and see why you should use it.
Ace Your Next Research Paper
Now, you are fully ready to nail your next research paper.
We know it can be a long and hard-working process, full of extensive research, critical thinking, and doubts.
But if you follow our step-by-step guide, you’ll see that it can be an interesting and simple experience.
Next time you have to do a research paper, you will ace it!
And if you are looking for more guidelines on how to improve your writing skills, don’t miss out on these related articles:

Argumentative Essay vs Narrative Essay | Everything You Need to Know

5 Common Essay Mistakes and How to Avoid Them

5 Hacks to Improve Your Creative Writing

How Much Does it Cost to Write My Essay?
- Best Apps and Tools
- Financial Tips and Scholarships
- Career Planning
- Non-Traditional Students
- Student Wellness
- Cost & Pricing

- 2525 Ponce de Leon Blvd Suite 300 Coral Gables, FL 33134 USA
- Phone: (786) 991-9293
- Gradehacker 2525 Ponce de Leon Blvd Suite 300 Coral Gables, FL 33134 USA
About Gradehacker
Business hours.
Mon - Fri: 10:00 am - 7 pm ET Sat - Sun: 10 am - 3 pm ET
© 2024 Gradehacker LLC All Rights Reserved.
Purdue Online Writing Lab Purdue OWL® College of Liberal Arts
Writing a Research Paper

Welcome to the Purdue OWL
This page is brought to you by the OWL at Purdue University. When printing this page, you must include the entire legal notice.
Copyright ©1995-2018 by The Writing Lab & The OWL at Purdue and Purdue University. All rights reserved. This material may not be published, reproduced, broadcast, rewritten, or redistributed without permission. Use of this site constitutes acceptance of our terms and conditions of fair use.
The Research Paper
There will come a time in most students' careers when they are assigned a research paper. Such an assignment often creates a great deal of unneeded anxiety in the student, which may result in procrastination and a feeling of confusion and inadequacy. This anxiety frequently stems from the fact that many students are unfamiliar and inexperienced with this genre of writing. Never fear—inexperience and unfamiliarity are situations you can change through practice! Writing a research paper is an essential aspect of academics and should not be avoided on account of one's anxiety. In fact, the process of writing a research paper can be one of the more rewarding experiences one may encounter in academics. What is more, many students will continue to do research throughout their careers, which is one of the reasons this topic is so important.
Becoming an experienced researcher and writer in any field or discipline takes a great deal of practice. There are few individuals for whom this process comes naturally. Remember, even the most seasoned academic veterans have had to learn how to write a research paper at some point in their career. Therefore, with diligence, organization, practice, a willingness to learn (and to make mistakes!), and, perhaps most important of all, patience, students will find that they can achieve great things through their research and writing.
The pages in this section cover the following topic areas related to the process of writing a research paper:
- Genre - This section will provide an overview for understanding the difference between an analytical and argumentative research paper.
- Choosing a Topic - This section will guide the student through the process of choosing topics, whether the topic be one that is assigned or one that the student chooses themselves.
- Identifying an Audience - This section will help the student understand the often times confusing topic of audience by offering some basic guidelines for the process.
- Where Do I Begin - This section concludes the handout by offering several links to resources at Purdue, and also provides an overview of the final stages of writing a research paper.

A step-by-step guide for creating and formatting APA Style student papers
The start of the semester is the perfect time to learn how to create and format APA Style student papers. This article walks through the formatting steps needed to create an APA Style student paper, starting with a basic setup that applies to the entire paper (margins, font, line spacing, paragraph alignment and indentation, and page headers). It then covers formatting for the major sections of a student paper: the title page, the text, tables and figures, and the reference list. Finally, it concludes by describing how to organize student papers and ways to improve their quality and presentation.
The guidelines for student paper setup are described and shown using annotated diagrams in the Student Paper Setup Guide (PDF, 3.40MB) and the A Step-by-Step Guide to APA Style Student Papers webinar . Chapter 1 of the Concise Guide to APA Style and Chapter 2 of the Publication Manual of the American Psychological Association describe the elements, format, and organization for student papers. Tables and figures are covered in Chapter 7 of both books. Information on paper format and tables and figures and a full sample student paper are also available on the APA Style website.
Basic setup
The guidelines for basic setup apply to the entire paper. Perform these steps when you first open your document, and then you do not have to worry about them again while writing your paper. Because these are general aspects of paper formatting, they apply to all APA Style papers, student or professional. Students should always check with their assigning instructor or institution for specific guidelines for their papers, which may be different than or in addition to APA Style guidelines.
Seventh edition APA Style was designed with modern word-processing programs in mind. Most default settings in programs such as Academic Writer, Microsoft Word, and Google Docs already comply with APA Style. This means that, for most paper elements, you do not have to make any changes to the default settings of your word-processing program. However, you may need to make a few adjustments before you begin writing.
Use 1-in. margins on all sides of the page (top, bottom, left, and right). This is usually how papers are automatically set.
Use a legible font. The default font of your word-processing program is acceptable. Many sans serif and serif fonts can be used in APA Style, including 11-point Calibri, 11-point Arial, 12-point Times New Roman, and 11-point Georgia. You can also use other fonts described on the font page of the website.
Line spacing
Double-space the entire paper including the title page, block quotations, and the reference list. This is something you usually must set using the paragraph function of your word-processing program. But once you do, you will not have to change the spacing for the entirety of your paper–just double-space everything. Do not add blank lines before or after headings. Do not add extra spacing between paragraphs. For paper sections with different line spacing, see the line spacing page.
Paragraph alignment and indentation
Align all paragraphs of text in the body of your paper to the left margin. Leave the right margin ragged. Do not use full justification. Indent the first line of every paragraph of text 0.5-in. using the tab key or the paragraph-formatting function of your word-processing program. For paper sections with different alignment and indentation, see the paragraph alignment and indentation page.
Page numbers
Put a page number in the top right of every page header , including the title page, starting with page number 1. Use the automatic page-numbering function of your word-processing program to insert the page number in the top right corner; do not type the page numbers manually. The page number is the same font and font size as the text of your paper. Student papers do not require a running head on any page, unless specifically requested by the instructor.
Title page setup
Title page elements.
APA Style has two title page formats: student and professional (for details, see title page setup ). Unless instructed otherwise, students should use the student title page format and include the following elements, in the order listed, on the title page:
- Paper title.
- Name of each author (also known as the byline).
- Affiliation for each author.
- Course number and name.
- Instructor name.
- Assignment due date.
- Page number 1 in the top right corner of the page header.
The format for the byline depends on whether the paper has one author, two authors, or three or more authors.
- When the paper has one author, write the name on its own line (e.g., Jasmine C. Hernandez).
- When the paper has two authors, write the names on the same line and separate them with the word “and” (e.g., Upton J. Wang and Natalia Dominguez).
- When the paper has three or more authors, separate the names with commas and include “and” before the final author’s name (e.g., Malia Mohamed, Jaylen T. Brown, and Nia L. Ball).
Students have an academic affiliation, which identities where they studied when the paper was written. Because students working together on a paper are usually in the same class, they will have one shared affiliation. The affiliation consists of the name of the department and the name of the college or university, separated by a comma (e.g., Department of Psychology, George Mason University). The department is that of the course to which the paper is being submitted, which may be different than the department of the student’s major. Do not include the location unless it is part of the institution’s name.
Write the course number and name and the instructor name as shown on institutional materials (e.g., the syllabus). The course number and name are often separated by a colon (e.g., PST-4510: History and Systems Psychology). Write the assignment due date in the month, date, and year format used in your country (e.g., Sept. 10, 2020).
Title page line spacing
Double-space the whole title page. Place the paper title three or four lines down from the top of the page. Add an extra double-spaced blank like between the paper title and the byline. Then, list the other title page elements on separate lines, without extra lines in between.
Title page alignment
Center all title page elements (except the right-aligned page number in the header).
Title page font
Write the title page using the same font and font size as the rest of your paper. Bold the paper title. Use standard font (i.e., no bold, no italics) for all other title page elements.
Text elements
Repeat the paper title at the top of the first page of text. Begin the paper with an introduction to provide background on the topic, cite related studies, and contextualize the paper. Use descriptive headings to identify other sections as needed (e.g., Method, Results, Discussion for quantitative research papers). Sections and headings vary depending on the paper type and its complexity. Text can include tables and figures, block quotations, headings, and footnotes.
Text line spacing
Double-space all text, including headings and section labels, paragraphs of text, and block quotations.
Text alignment
Center the paper title on the first line of the text. Indent the first line of all paragraphs 0.5-in.
Left-align the text. Leave the right margin ragged.
Block quotation alignment
Indent the whole block quotation 0.5-in. from the left margin. Double-space the block quotation, the same as other body text. Find more information on the quotations page.
Use the same font throughout the entire paper. Write body text in standard (nonbold, nonitalic) font. Bold only headings and section labels. Use italics sparingly, for instance, to highlight a key term on first use (for more information, see the italics page).
Headings format
For detailed guidance on formatting headings, including headings in the introduction of a paper, see the headings page and the headings in sample papers .
- Alignment: Center Level 1 headings. Left-align Level 2 and Level 3 headings. Indent Level 4 and Level 5 headings like a regular paragraph.
- Font: Boldface all headings. Also italicize Level 3 and Level 5 headings. Create heading styles using your word-processing program (built into AcademicWriter, available for Word via the sample papers on the APA Style website).
Tables and figures setup
Tables and figures are only included in student papers if needed for the assignment. Tables and figures share the same elements and layout. See the website for sample tables and sample figures .
Table elements
Tables include the following four elements:
- Body (rows and columns)
- Note (optional if needed to explain elements in the table)
Figure elements
Figures include the following four elements:
- Image (chart, graph, etc.)
- Note (optional if needed to explain elements in the figure)
Table line spacing
Double-space the table number and title. Single-, 1.5-, or double-space the table body (adjust as needed for readability). Double-space the table note.
Figure line spacing
Double-space the figure number and title. The default settings for spacing in figure images is usually acceptable (but adjust the spacing as needed for readability). Double-space the figure note.
Table alignment
Left-align the table number and title. Center column headings. Left-align the table itself and left-align the leftmost (stub) column. Center data in the table body if it is short or left-align the data if it is long. Left-align the table note.
Figure alignment
Left-align the figure number and title. Left-align the whole figure image. The default alignment of the program in which you created your figure is usually acceptable for axis titles and data labels. Left-align the figure note.
Bold the table number. Italicize the table title. Use the same font and font size in the table body as the text of your paper. Italicize the word “Note” at the start of the table note. Write the note in the same font and font size as the text of your paper.
Figure font
Bold the figure number. Italicize the figure title. Use a sans serif font (e.g., Calibri, Arial) in the figure image in a size between 8 to 14 points. Italicize the word “Note” at the start of the figure note. Write the note in the same font and font size as the text of your paper.
Placement of tables and figures
There are two options for the placement of tables and figures in an APA Style paper. The first option is to place all tables and figures on separate pages after the reference list. The second option is to embed each table and figure within the text after its first callout. This guide describes options for the placement of tables and figures embedded in the text. If your instructor requires tables and figures to be placed at the end of the paper, see the table and figure guidelines and the sample professional paper .
Call out (mention) the table or figure in the text before embedding it (e.g., write “see Figure 1” or “Table 1 presents”). You can place the table or figure after the callout either at the bottom of the page, at the top of the next page, or by itself on the next page. Avoid placing tables and figures in the middle of the page.
Embedding at the bottom of the page
Include a callout to the table or figure in the text before that table or figure. Add a blank double-spaced line between the text and the table or figure at the bottom of the page.
Embedding at the top of the page
Include a callout to the table in the text on the previous page before that table or figure. The table or figure then appears at the top of the next page. Add a blank double-spaced line between the end of the table or figure and the text that follows.
Embedding on its own page
Embed long tables or large figures on their own page if needed. The text continues on the next page.
Reference list setup
Reference list elements.
The reference list consists of the “References” section label and the alphabetical list of references. View reference examples on the APA Style website. Consult Chapter 10 in both the Concise Guide and Publication Manual for even more examples.
Reference list line spacing
Start the reference list at the top of a new page after the text. Double-space the entire reference list (both within and between entries).
Reference list alignment
Center the “References” label. Apply a hanging indent of 0.5-in. to all reference list entries. Create the hanging indent using your word-processing program; do not manually hit the enter and tab keys.
Reference list font
Bold the “References” label at the top of the first page of references. Use italics within reference list entries on either the title (e.g., webpages, books, reports) or on the source (e.g., journal articles, edited book chapters).
Final checks
Check page order.
- Start each section on a new page.
- Arrange pages in the following order:
- Title page (page 1).
- Text (starts on page 2).
- Reference list (starts on a new page after the text).
Check headings
- Check that headings accurately reflect the content in each section.
- Start each main section with a Level 1 heading.
- Use Level 2 headings for subsections of the introduction.
- Use the same level of heading for sections of equal importance.
- Avoid having only one subsection within a section (have two or more, or none).
Check assignment instructions
- Remember that instructors’ guidelines supersede APA Style.
- Students should check their assignment guidelines or rubric for specific content to include in their papers and to make sure they are meeting assignment requirements.
Tips for better writing
- Ask for feedback on your paper from a classmate, writing center tutor, or instructor.
- Budget time to implement suggestions.
- Use spell-check and grammar-check to identify potential errors, and then manually check those flagged.
- Proofread the paper by reading it slowly and carefully aloud to yourself.
- Consult your university writing center if you need extra help.
About the author

Undergraduate student resources
- Search Menu
- Sign in through your institution
- Advance articles
- Editor's Choice
- Supplements
- French Abstracts
- Portuguese Abstracts
- Spanish Abstracts
- Author Guidelines
- Submission Site
- Open Access
- About International Journal for Quality in Health Care
- About the International Society for Quality in Health Care
- Editorial Board
- Advertising and Corporate Services
- Journals Career Network
- Self-Archiving Policy
- Dispatch Dates
- Contact ISQua
- Journals on Oxford Academic
- Books on Oxford Academic

Article Contents
Primacy of the research question, structure of the paper, writing a research article: advice to beginners.
- Article contents
- Figures & tables
- Supplementary Data
Thomas V. Perneger, Patricia M. Hudelson, Writing a research article: advice to beginners, International Journal for Quality in Health Care , Volume 16, Issue 3, June 2004, Pages 191–192, https://doi.org/10.1093/intqhc/mzh053
- Permissions Icon Permissions
Writing research papers does not come naturally to most of us. The typical research paper is a highly codified rhetorical form [ 1 , 2 ]. Knowledge of the rules—some explicit, others implied—goes a long way toward writing a paper that will get accepted in a peer-reviewed journal.
A good research paper addresses a specific research question. The research question—or study objective or main research hypothesis—is the central organizing principle of the paper. Whatever relates to the research question belongs in the paper; the rest doesn’t. This is perhaps obvious when the paper reports on a well planned research project. However, in applied domains such as quality improvement, some papers are written based on projects that were undertaken for operational reasons, and not with the primary aim of producing new knowledge. In such cases, authors should define the main research question a posteriori and design the paper around it.
Generally, only one main research question should be addressed in a paper (secondary but related questions are allowed). If a project allows you to explore several distinct research questions, write several papers. For instance, if you measured the impact of obtaining written consent on patient satisfaction at a specialized clinic using a newly developed questionnaire, you may want to write one paper on the questionnaire development and validation, and another on the impact of the intervention. The idea is not to split results into ‘least publishable units’, a practice that is rightly decried, but rather into ‘optimally publishable units’.
What is a good research question? The key attributes are: (i) specificity; (ii) originality or novelty; and (iii) general relevance to a broad scientific community. The research question should be precise and not merely identify a general area of inquiry. It can often (but not always) be expressed in terms of a possible association between X and Y in a population Z, for example ‘we examined whether providing patients about to be discharged from the hospital with written information about their medications would improve their compliance with the treatment 1 month later’. A study does not necessarily have to break completely new ground, but it should extend previous knowledge in a useful way, or alternatively refute existing knowledge. Finally, the question should be of interest to others who work in the same scientific area. The latter requirement is more challenging for those who work in applied science than for basic scientists. While it may safely be assumed that the human genome is the same worldwide, whether the results of a local quality improvement project have wider relevance requires careful consideration and argument.
Once the research question is clearly defined, writing the paper becomes considerably easier. The paper will ask the question, then answer it. The key to successful scientific writing is getting the structure of the paper right. The basic structure of a typical research paper is the sequence of Introduction, Methods, Results, and Discussion (sometimes abbreviated as IMRAD). Each section addresses a different objective. The authors state: (i) the problem they intend to address—in other terms, the research question—in the Introduction; (ii) what they did to answer the question in the Methods section; (iii) what they observed in the Results section; and (iv) what they think the results mean in the Discussion.
In turn, each basic section addresses several topics, and may be divided into subsections (Table 1 ). In the Introduction, the authors should explain the rationale and background to the study. What is the research question, and why is it important to ask it? While it is neither necessary nor desirable to provide a full-blown review of the literature as a prelude to the study, it is helpful to situate the study within some larger field of enquiry. The research question should always be spelled out, and not merely left for the reader to guess.
Typical structure of a research paper
| Introduction |
| State why the problem you address is important |
| State what is lacking in the current knowledge |
| State the objectives of your study or the research question |
| Methods |
| Describe the context and setting of the study |
| Specify the study design |
| Describe the ‘population’ (patients, doctors, hospitals, etc.) |
| Describe the sampling strategy |
| Describe the intervention (if applicable) |
| Identify the main study variables |
| Describe data collection instruments and procedures |
| Outline analysis methods |
| Results |
| Report on data collection and recruitment (response rates, etc.) |
| Describe participants (demographic, clinical condition, etc.) |
| Present key findings with respect to the central research question |
| Present secondary findings (secondary outcomes, subgroup analyses, etc.) |
| Discussion |
| State the main findings of the study |
| Discuss the main results with reference to previous research |
| Discuss policy and practice implications of the results |
| Analyse the strengths and limitations of the study |
| Offer perspectives for future work |
| Introduction |
| State why the problem you address is important |
| State what is lacking in the current knowledge |
| State the objectives of your study or the research question |
| Methods |
| Describe the context and setting of the study |
| Specify the study design |
| Describe the ‘population’ (patients, doctors, hospitals, etc.) |
| Describe the sampling strategy |
| Describe the intervention (if applicable) |
| Identify the main study variables |
| Describe data collection instruments and procedures |
| Outline analysis methods |
| Results |
| Report on data collection and recruitment (response rates, etc.) |
| Describe participants (demographic, clinical condition, etc.) |
| Present key findings with respect to the central research question |
| Present secondary findings (secondary outcomes, subgroup analyses, etc.) |
| Discussion |
| State the main findings of the study |
| Discuss the main results with reference to previous research |
| Discuss policy and practice implications of the results |
| Analyse the strengths and limitations of the study |
| Offer perspectives for future work |
The Methods section should provide the readers with sufficient detail about the study methods to be able to reproduce the study if so desired. Thus, this section should be specific, concrete, technical, and fairly detailed. The study setting, the sampling strategy used, instruments, data collection methods, and analysis strategies should be described. In the case of qualitative research studies, it is also useful to tell the reader which research tradition the study utilizes and to link the choice of methodological strategies with the research goals [ 3 ].
The Results section is typically fairly straightforward and factual. All results that relate to the research question should be given in detail, including simple counts and percentages. Resist the temptation to demonstrate analytic ability and the richness of the dataset by providing numerous tables of non-essential results.
The Discussion section allows the most freedom. This is why the Discussion is the most difficult to write, and is often the weakest part of a paper. Structured Discussion sections have been proposed by some journal editors [ 4 ]. While strict adherence to such rules may not be necessary, following a plan such as that proposed in Table 1 may help the novice writer stay on track.
References should be used wisely. Key assertions should be referenced, as well as the methods and instruments used. However, unless the paper is a comprehensive review of a topic, there is no need to be exhaustive. Also, references to unpublished work, to documents in the grey literature (technical reports), or to any source that the reader will have difficulty finding or understanding should be avoided.
Having the structure of the paper in place is a good start. However, there are many details that have to be attended to while writing. An obvious recommendation is to read, and follow, the instructions to authors published by the journal (typically found on the journal’s website). Another concerns non-native writers of English: do have a native speaker edit the manuscript. A paper usually goes through several drafts before it is submitted. When revising a paper, it is useful to keep an eye out for the most common mistakes (Table 2 ). If you avoid all those, your paper should be in good shape.
Common mistakes seen in manuscripts submitted to this journal
| The research question is not specified |
| The stated aim of the paper is tautological (e.g. ‘The aim of this paper is to describe what we did’) or vague (e.g. ‘We explored issues related to X’) |
| The structure of the paper is chaotic (e.g. methods are described in the Results section) |
| The manuscripts does not follow the journal’s instructions for authors |
| The paper much exceeds the maximum number of words allowed |
| The Introduction is an extensive review of the literature |
| Methods, interventions and instruments are not described in sufficient detail |
| Results are reported selectively (e.g. percentages without frequencies, -values without measures of effect) |
| The same results appear both in a table and in the text |
| Detailed tables are provided for results that do not relate to the main research question |
| In the Introduction and Discussion, key arguments are not backed up by appropriate references |
| References are out of date or cannot be accessed by most readers |
| The Discussion does not provide an answer to the research question |
| The Discussion overstates the implications of the results and does not acknowledge the limitations of the study |
| The paper is written in poor English |
| The research question is not specified |
| The stated aim of the paper is tautological (e.g. ‘The aim of this paper is to describe what we did’) or vague (e.g. ‘We explored issues related to X’) |
| The structure of the paper is chaotic (e.g. methods are described in the Results section) |
| The manuscripts does not follow the journal’s instructions for authors |
| The paper much exceeds the maximum number of words allowed |
| The Introduction is an extensive review of the literature |
| Methods, interventions and instruments are not described in sufficient detail |
| Results are reported selectively (e.g. percentages without frequencies, -values without measures of effect) |
| The same results appear both in a table and in the text |
| Detailed tables are provided for results that do not relate to the main research question |
| In the Introduction and Discussion, key arguments are not backed up by appropriate references |
| References are out of date or cannot be accessed by most readers |
| The Discussion does not provide an answer to the research question |
| The Discussion overstates the implications of the results and does not acknowledge the limitations of the study |
| The paper is written in poor English |
Huth EJ . How to Write and Publish Papers in the Medical Sciences , 2nd edition. Baltimore, MD: Williams & Wilkins, 1990 .
Browner WS . Publishing and Presenting Clinical Research . Baltimore, MD: Lippincott, Williams & Wilkins, 1999 .
Devers KJ , Frankel RM. Getting qualitative research published. Educ Health 2001 ; 14 : 109 –117.
Docherty M , Smith R. The case for structuring the discussion of scientific papers. Br Med J 1999 ; 318 : 1224 –1225.
| Month: | Total Views: |
|---|---|
| December 2016 | 1 |
| January 2017 | 242 |
| February 2017 | 451 |
| March 2017 | 632 |
| April 2017 | 289 |
| May 2017 | 349 |
| June 2017 | 347 |
| July 2017 | 752 |
| August 2017 | 649 |
| September 2017 | 844 |
| October 2017 | 920 |
| November 2017 | 1,646 |
| December 2017 | 7,530 |
| January 2018 | 8,339 |
| February 2018 | 9,141 |
| March 2018 | 13,810 |
| April 2018 | 19,070 |
| May 2018 | 16,599 |
| June 2018 | 13,752 |
| July 2018 | 12,558 |
| August 2018 | 15,395 |
| September 2018 | 14,283 |
| October 2018 | 14,089 |
| November 2018 | 17,418 |
| December 2018 | 16,718 |
| January 2019 | 17,941 |
| February 2019 | 15,452 |
| March 2019 | 17,862 |
| April 2019 | 18,214 |
| May 2019 | 17,643 |
| June 2019 | 13,983 |
| July 2019 | 13,079 |
| August 2019 | 12,840 |
| September 2019 | 12,724 |
| October 2019 | 10,555 |
| November 2019 | 9,256 |
| December 2019 | 7,084 |
| January 2020 | 7,476 |
| February 2020 | 8,890 |
| March 2020 | 8,359 |
| April 2020 | 13,466 |
| May 2020 | 6,115 |
| June 2020 | 8,233 |
| July 2020 | 7,063 |
| August 2020 | 6,487 |
| September 2020 | 8,284 |
| October 2020 | 9,266 |
| November 2020 | 10,248 |
| December 2020 | 10,201 |
| January 2021 | 9,786 |
| February 2021 | 10,582 |
| March 2021 | 10,011 |
| April 2021 | 10,238 |
| May 2021 | 9,880 |
| June 2021 | 8,729 |
| July 2021 | 6,278 |
| August 2021 | 6,723 |
| September 2021 | 7,704 |
| October 2021 | 8,604 |
| November 2021 | 9,733 |
| December 2021 | 7,678 |
| January 2022 | 7,286 |
| February 2022 | 7,406 |
| March 2022 | 8,097 |
| April 2022 | 7,589 |
| May 2022 | 8,337 |
| June 2022 | 5,305 |
| July 2022 | 3,959 |
| August 2022 | 4,166 |
| September 2022 | 5,435 |
| October 2022 | 5,294 |
| November 2022 | 5,096 |
| December 2022 | 4,104 |
| January 2023 | 3,550 |
| February 2023 | 4,079 |
| March 2023 | 4,935 |
| April 2023 | 3,793 |
| May 2023 | 3,689 |
| June 2023 | 2,548 |
| July 2023 | 2,313 |
| August 2023 | 2,125 |
| September 2023 | 2,172 |
| October 2023 | 2,859 |
| November 2023 | 2,767 |
| December 2023 | 2,335 |
| January 2024 | 2,825 |
| February 2024 | 2,630 |
| March 2024 | 2,874 |
| April 2024 | 2,311 |
| May 2024 | 2,108 |
| June 2024 | 1,586 |
| July 2024 | 8,045 |
| August 2024 | 2,672 |
| September 2024 | 1,312 |
Email alerts
Citing articles via.
- Recommend to your Library
Affiliations
- Online ISSN 1464-3677
- Print ISSN 1353-4505
- Copyright © 2024 International Society for Quality in Health Care and Oxford University Press
- About Oxford Academic
- Publish journals with us
- University press partners
- What we publish
- New features
- Open access
- Institutional account management
- Rights and permissions
- Get help with access
- Accessibility
- Advertising
- Media enquiries
- Oxford University Press
- Oxford Languages
- University of Oxford
Oxford University Press is a department of the University of Oxford. It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide
- Copyright © 2024 Oxford University Press
- Cookie settings
- Cookie policy
- Privacy policy
- Legal notice
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This PDF is available to Subscribers Only
For full access to this pdf, sign in to an existing account, or purchase an annual subscription.
We use cookies to give you the best experience possible. By continuing we’ll assume you’re on board with our cookie policy

- A Research Guide
- Research Paper Guide
How to Write a Research Paper
- STEP 1. How to start research topic?
- STEP 2. Find information
- STEP 3. Make your thesis statement
- STEP 4. Make research paper outline
- STEP 5. Oganize your notes
- STEP 6. Literature review
STEP 7. The research question(s)
Step 8. research methodology.
- STEP 9. Writing the results, analysis, discussion, and conclusion
STEP 10. The process of writing a research paper
- STEP 11. Write your first draft
- Checklist One
- Checklist Two
- STEP 13. Tools for research paper help
- STEP 14. Some words of encouragement
When an average student hears about writing a research paper, apprehension and anxiety always arise. Contrary to popular belief, it’s easier than writing an average college assignment. Essentially, what is a research paper in basic terms? It is a type of academic writing that must follow instructions to the letter and adhere to certain standards. Since writing a research paper is based on the original research already done by another person, your task involves analysis, processing, and interpretation of research outcomes.
Since it’s a time-consuming process, it’s only natural to feel stressed without clear knowledge regarding where to start. If you write a research paper for the first time, things can become even more difficult. As students need to learn more about research paper structure peculiarities, they often write hastily and skip through all the essential elements that make research papers accurate.
This helpful guide will provide advice and assistance as you navigate the challenges of research paper writing. The article on how to write a research paper will include all the recommended steps that must be followed to earn the best grades. We shall also provide helpful tips and 14 steps to help you choose a good topic, write without stress and collect all the necessary information. It is precisely what helps to start up research work and achieve success.
You will also learn more about citation basics and free online tools that can help you check certain things and achieve clarity in your tone and the writing flow as you write. Read on to learn how to compose an A+ research paper!
STEP 1. How to start research and find a good topic?
The most challenging part is knowing how to start a research paper by narrowing things down. You must start by choosing a subject that interests and motivates you the most. Your motivation and general attitude will always differ when you feel inspired by the research problem. It will also help determine things you might already know regarding the subject.
Stay focused on keeping your content narrowed down. If you are talking about social conflicts, specify what types of problems it represents by talking about location and the sample group. When unsure, always talk to your academic advisor and seek topic approval first to ensure you are on the right track. It will always help you to avoid confusion in the future!
Finding a suitable topic problem or how to make your paper stand out!
When you have several research topics that inspire you, focus on those you know better and think about subjects that can be supported with solid argumentative evidence. Invest your time in a preliminary study of the subject and pay attention to detail because it will help you to see the shortcomings and advantages of choosing a particular subject.
Once you can achieve this goal for your paper, you will also meet most grading rubric requirements. As you choose your research paper writing topic, try to determine research questions early, as it will help you to see how to work with the methods section, analysis, and discussion, among other vital elements. Make sure you look at similar research paper topics by checking what else is available online.
This type of exploration is always essential because you must make your research paper stand out from the rest by offering something unique. You must do relevant explorations to see what other authors have done so you do not repeat the same work. Outline the objectives you wish to follow and develop the thesis or a hypothesis for your future work. Ask yourself what kind of work has already been done as you focus on your chosen topic.
Ask yourself whether some bits and pieces have yet to be explored. This way, you can shed some light on a subject and provide a new research method that has yet to be explored. Moreover, it will help you to publish your work in the future and present it at various academic venues! When your professor sees that you have done your homework correctly, it will become a cornerstone in your academic career!
Narrowing things down!
The most common mistake most college students make is taking a narrow approach when starting with research work. The trick here is to narrow things down and focus on the vital information you have obtained. Consider the statistical data, literature reviews, and facts supporting your main arguments. It means you do not have to choose all the available information and write it down in your work. Such an approach will only make your paper sound generic and take the research part away.
Think about choosing another method by focusing on elements that interest you the most! Seek the value in every sentence and add your author’s voice as you process information and present something of your vision. It will also help to explain why your research paper is important as you talk about the significance of your work.
STEP 2. Finding credible sources of information
One of the first elements of proper research work is finding the information required for an outline. Consider finding relevant general information online by visiting libraries or university resources. You may also use search engines and approach online resources such as Britannica . Still, try to implement Google Scholar as well by checking relevant publications.
It will be a good starting point to consider! Ensure you look at the domain extensions representing educational institutions, the .edu or .org domains (non-profit organizations). Likewise, you can locate accurate and verified information at .gov extension websites. Just remember that there may be a certain political bias, depending on the government.
Be careful with commercial .com websites as you write research papers. Many such websites can be excellent for research purposes, yet many tend to contain poor quality or advertisements that may make them less relevant. You can look at Network Solutions websites to learn more about what extensions this or that type stand for. Quality sources are not easy to find, so you must learn how to evaluate websites critically and eliminate those sources that are not peer-reviewed. If you are looking for sources in print or seek digital books for your academic paper, consider checking the Online Public Access Catalog (OPAC) library.
Conduct preliminary research based on a chosen subject!
When you are visiting a library online to write a research paper or considering visiting one in person, make sure to check the following as you write and take notes:
- Almanacs, atlases, and scientific catalogs;
- Government publications, guides, and reports;
- Vertical files;
- Encyclopedias and Dictionaries;
- Magazines and Newspapers;
- Yellow Pages.
Speaking of online resources, you can safely consider web-based information as long as you can check the original or consider multimedia sources in audio and/or video format:
- Online reference materials at SIRS, ProQuest, or eLibrary;
- Indexing is done for periodicals and newspapers (onlinenewspapers.com);
- Newspapers and scientific magazines;
- Answers.com;
- International Public Libraries;
- Wall Street Executive Library;
- Online Encyclopedias like Britannica or Canadian Encyclopedia;
- Google Scholar.
Remember to check public and university libraries, read business press releases related to your subject, or check governmental agencies as you write. You can also talk to people by hosting interviews for the primary sources. Always document your sources, and do not use anything without a solid reason. When collecting your research paper writing resources, take notes of all the citing and/or bibliographical information.
It must include the author’s name, full title, place of publication, information about the publisher, publication date, and page numbers. Include the URL, DOI, or ISBN, depending on the publication’s type and the paper type you must write. As a rule, information not providing sufficient bibliographical data is virtually useless for citation purposes. If you are unsure of some source, it’s better to avoid it.
Try the new AI Essay Writer to make writing easier. It’s free and a great solution for moments when creativity seems out of reach!
STEP 3. Make your thesis statement clear
A thesis statement is the heart of every research paper because they deliver the main thesis or an assumption that is made. As a rule, the thesis must be created when you work on your paper’s outline. While some research papers may not require a thesis, the list of objectives must be presented in any case to help your target audience understand what they might expect. A thesis is the main idea of your research and a central point that should be outlined after your introduction part. The arguments must be clear and related to your chosen subject and topic.
In most cases, a thesis statement should present one sentence only. You can approach it as a declaration of your vision and the message that you wish to deliver. This is how you defend your point!
Developing a thesis statement before you do all the rest is recommended. A thesis usually appears at the end of your introduction paragraph and can be placed in italics. Starting with your thesis statement when you already have certain arguments written down is not recommended, as it will require post-adjustment work.
Likewise, your thesis statement must be supported by the evidence and literature sources you have obtained. Take your time to analyze available materials and come up with a clearly formulated statement for your research paper. It will help you to develop your ideas further as you move to the body parts of your research.
Do your best to avoid generic or vague statements and always focus on the subject and keywords included in your main subject. Your paper should inspire the audience and reflect the main idea presented in your introduction. Remember to avoid placing citations in the same paragraph with your thesis. It is precisely where your unique ideas and vision must be offered instead!
Your thesis statement should achieve the following as you write:
- Outline and explain how you approach a particular research subject.
- Address the research questions you have been given to write a research paper.
- Explain what to expect from your research work.
- Present various claims and set up a dispute.
Your thesis statement should be valid and possible to achieve. It is recommended to share it with your academic advisor as you may receive brief revision comments and address all the weak points. If it’s impossible, proofread it aloud and check if it can be supported.
Here is a list of questions to consider as you write a scientific paper:
- Does my thesis address the main problem of my research paper?
- Can I support my thesis with sufficient evidence?
- Does my thesis provide interesting and inspiring data?
- Is my statement clear and precise to present academic value?
- Can my thesis position be disputed and challenged? As a rule, the answer should be “yes”.
Remember that changing your thesis’s wording is possible as you work on your assignment. It will help you shape better ideas and increase your precision. Speaking of helpful research paper tips based on writing a powerful paper, you should focus on the credibility of your writing by checking things twice. Your strong and clear thesis shows that you have done your homework correctly and know the subject well!
STEP 4. Creating a research paper outline
Let’s continue with the basic research paper outline template that can be used further for research paper writing purposes. Here is the research paper outline format with universal elements:
I. INTRODUCTION
a) Provide a brief overview of a problem or issue you plan to research. Include your main assumption or an argument outlined in your thesis statement.
b) Include a justification of your research work. It is basically a reason why your readers should care and follow your research paper. Also known as the study importance, you must write it clearly and explain why your subject is meaningful.
c) Write down a brief outline of the paper’s range and the planned methods you will use to approach your issue or problem.
II. RESEARCH PROBLEM
a) Provide a background history of a problem.
b) How does your issue impact society and/or the academic environment?
c) Critical factors related to your research problem.
d) Possible solutions that will be explored in your paper.
III. LITERATURE REVIEW
a) A list of theories and concepts (textbooks, journal articles, or other relevant publications.
i. Offer a description of how these theories help to explain your problem and represent a solution.
ii. How were these concepts of theories explained by others?
iii. Describe how these theories help to explain your research problem.
b) Empirical research literature (mostly journal articles)
i. Provide an overview of relevant empirical studies based on chronology.
ii. Offer a summary of the methodology.
iii. Talk about major findings.
iv. What were the limitations you faced?
c) Talk about what has been discovered in a literature review
i. Talk about concepts and definitions you plan to use based on other authors.
ii. Describe all the unique concepts you have faced.
iii. Describe what method fits your research best and based on what reading(s).
a. Offer specific research questions that you address.
b. Describe your research method and data collection processes.
c. Justify your method’s rationale and explain why you have chosen it.
V. RESULTS AND DISCUSSION
a. Write down your major findings.
b. Write about statistical information and facts to explain your research outcomes.
c. Discuss and write about the relevance of findings based on prior studies.
d. Was there anything unusual or not-so-common? Write about unusual discoveries.
e. The discovered limitations of your study as you wrote a research paper.
VI. CONCLUSION AND RECOMMENDATIONS
a. Provide a brief overview of the issue examined and outline major findings.
b. Remind your readers (briefly) about the goals of your study and write down your accomplishments.
c. Offer recommendations and talk about how other researchers can benefit from your findings.
Depending on the type of research you have to write, you can write a scientific paper with an outline that can be formal or informal. The authors use an informal outline to narrow down and organize their ideas. You can revise, add, edit, or remove certain bits mentioned in your paper without keeping it strictly focused. You can use it as a writer who wants to keep on track.
Now, things are totally different when you are asked to present a formal outline and research paper. A formal outline always uses a clear structure by following numbers, letters, and logic. Every paper heading is essential here, along with sub-headings that must be grouped exactly as they appear in your research paper. The capital Roman numerals are usually used for coordination purposes.
An example of an outline to help you write a research paper:
Paper Title: Open Scouting Movement in Flanders, Belgium. An Analysis of Scouting Branches
a. Importance of Open Scouting Movement in Belgium.
b. Define the major differences between Open Scouting and the National Scouting Movement.
c. A brief history of Open Scouting in Belgium.
d. Examination of statistical data to determine the involvement of scouting branches.
e) Justification of the study’s importance.
II. LITERATURE REVIEW
a. Analysis of popular theories in youth movements in Belgium: Chiro, KSA, and Vrije-school movements.
b. Review of youth studies done based on local coverage and accessibility.
c. Gaps and areas that lack coverage and discussion in literature and the media.
III. METHOD
a. Visiting Open Scouting branches in person.
b. Behavior codes across branches to compare the rules.
c. Data collection methods: staff surveys and group interviews.
IV. RESULTS, DISCUSSION, AND ANALYSIS
a. Differences and similarities of scouting branches review.
b. Analysis of practical differences of Open Scouting in Belgium (write a research paper based on observations).
c. Outcomes of surveys.
d. Usefulness and accuracy of relevant methods (write research paper points related to your methodology).
e. Limitations of the implemented methods.
V. CONCLUSION AND RECOMMENDATION
a. Main points mastered by the research.
b. The importance of Open Scouting in practice.
c. Further recommendations for parents, educators, and camp leaders.
As you do a research paper, it is recommended to narrow things down and keep your outline to the point based on what data you have obtained for your paper. When you have an outline, you can avoid being vague and keep your structure relevant to a certain logic. When you have a good outline and write a research paper correctly, it is essential to write an excellent one. Remember to follow our templates and that an outline should include an introduction, body content, and a conclusion.
Writing an introduction and other elements for your research paper structure
- INTRODUCTION: This is where you have to introduce your research problem and include a clear thesis. Ask yourself about the main reason why you are researching something and outline it here. Explain how you plan to approach an issue in your research paper. Depending on the research type, you can focus on the facts, review relevant literature, or offer an analysis of a problem. This is also where you must explain all the major points mentioned in your paper and why they matter!
- BODY CONTENT: Present your strongest to weakest arguments by supporting your thesis statement. This section is also divided into several parts. It will always depend on your academic discipline, requirements issued by your professors, subject, and many other things. To write a good research paper, your body parts usually contain elements like a literature review, methodology or methods section, analysis, research outcomes (results), sample analysis, and a discussion section.
- A CONCLUSION: This is where you must state your thesis and research question in simpler terms. Do not introduce any new ideas. Try your best to summarize existing arguments. Explain why you came up with a specific conclusion. Your paper must represent at least 15% of the final part. Explain again why your research matters and how the results can be replicated or used for future research.
STEP 5. Organize your notes
Keeping your notes organized is essential as you have to ensure that every vital point has been mentioned as it is presented in your outline. As you write a research paper, evaluate your research data critically and check for accuracy. If some information has not been updated in a while and has missing citation data, it’s always better to skip it as you work on your paper. If some beliefs or views oppose your main thesis, write a research essay section that mentions them in a counter-arguments paragraph.
It is one of the most important stages to consider when writing a research paper. It is where you must sort, analyze, and evaluate your available data. It will help you to learn new things and determine the main purpose of your objectives. Think about eliminating less important aspects as you take notes of your ideas, thoughts, concepts, and research findings.
As you write a research paper, avoid information that is not relevant to your research issue! Do not include facts that cannot be supported by a clear piece of evidence or a direct citation. At the same time, using only up to three citations per 300 words is recommended to avoid possible plagiarism risks. As you write a research paper, use paraphrasing instead whenever possible! If an idea is not yours, always provide a reference and document it accurately.
When you sort and classify your notes as you make a research paper stand out, do your best to provide detailed bibliographic data for each citation before you create a References or Works Cited page. As you work on the notes, you have prepared to write a research paper, use different outline codes or colors to mark the types of sources, depending if they are in print or online, related to the first, second, third argument, and so on.
STEP 6. A literature review or learning of available resources
This part basically stands for knowing what data is available out there. It is precisely where you have to do all the research work and a reason why it differs from most other assignment types you might encounter as a college or university student. The purpose here is not simply to list all the information available on a particular subject but to contribute something based on your vision and thoughts.
One of the ways to do that is to go through the literature review process and narrow things down as you look for background information related to a particular subject. As you start with a literature review for your paper, you automatically determine what is already known about an issue you plan to explore. A comprehensive literature review for a professional research paper will help you save time and discover what aspects of a subject have yet to be reviewed. Likewise, you will know what has been done before.
- Internet research based on keywords. It’s one of the easiest methods to start with as you look for information online and use specific keywords that reflect your chosen topic. You can consider the information available on various websites or general publications. Most students who write research papers will turn to academic research and scientific databases. Finding peer-reviewed sources and sorting out unnecessary social media websites or blogs is safe. Remember that places like PubMed or ScienceDirect are more trustworthy for an academic research paper than a post on Twitter, even though the latter may represent a primary source.
- Checking prior research on the topic. The next step worth taking is exploring all the prior work that other researchers have done. You can visit official organizations that work in the field of your research. This way, you can collect statistical information and see what they have found before and what information has been made available. Focus on whether research work is funded publicly or done privately. If the research is affiliated with a certain company, consider checking things twice for possible bias, especially if your paper must be neutral. Always check for credibility and try to locate information about the author(s)
- Visiting the university library. As a rule, if you are enrolled in Automotive Industry at MIT, you will have access to unique materials at the local library. The chances are high that your academic advisor may already provide you with a recommended or even obligatory reading list. As you brainstorm various steps in writing a research paper, take your time to research what’s available at the university or college library. Turn to research databases and look through the online index by entering keywords related to your paper. Most of them will have available citation info to save you valuable time.
- Using academic sources. These include peer-reviewed journals that you can access either in print or online. As a rule, such publications receive the highest level of credibility from most professors and should be considered first. It is partially because of the unbiased review and cross-linked publications. If you are dealing with a citation from someone who knows the subject well, you will increase the credibility of your research paper. Likewise, if your research paper becomes published or you present it publicly, you increase your chances of being cited and quoted by others in the chosen scientific field.
Once you complete a detailed literature review and are ready to pursue the next steps to write a research paper, you will receive sufficient background information to understand your subject’s peculiarities. Much of this work will help you shape your research paper thesis much better because you will automatically address numerous objectives and know all the limitations and research gaps. You will learn what must be done to keep the research accessible and clear based on the chosen topic.
Before moving to the Methodology section, exploring the answers that address your research objectives is crucial. Most importantly, you must keep your research paper data within the scope and timeframe of the issue. When dealing with scientific research, you must keep information clear to a certain point. It must be possible to replicate and measure your actions. Whether using quantitative or qualitative methods (or both), it should be trustworthy and possible to replicate.
Always consider what methods to use and what sources to put forward, depending on your subject. If you are talking about the violence of video games and their effect on teenagers, the best solution to write such a paper would be to use surveys or other methods in your research paper guidelines to determine your sampling method and avoid bias. Likewise, you may use various means and technical tools to receive answers to your research questions , depending on the strengths and weaknesses of each.
It’s one of the most challenging steps to consider as you have to choose a particular research method based on your research paper subject, the type of research, and the sources you have obtained. Here are the most popular research methods that you may consider:
- Focus group. It can be a suitable method if the author’s goal is to obtain information from a small group of people. It might be a safe choice for your research paper if you cannot invest time and funds. It usually comes to asking questions and taking notes as you determine a limited sample group. While it can be convenient, findings from a focus group method might not fit those cases where you need to be more careful with a selection. It means that you must avoid it for legal or medical studies. After all, as you write a research paper based on that, a fellow researcher can make a limited conclusion regarding the findings because of the limited sampling approach.
- Survey. It’s another popular method that can be used for a large sample group where participants are chosen randomly. Such a method is often recommended when dealing with peer-reviewed research sources. When writing a scientific paper, remember that most paper surveys you can write down also have weaknesses since participants may not provide honest opinions or politically correct answers. Numerous factors also influence them, so a certain bias is always possible.
- Field experiments . These are also helpful when you wish to experience information in practice and explore a certain community. The methods used here include field analysis, lab experiments, direct observation, participant control, case study replication, and many non-conventional methods that researchers can use to address their research objectives. The same is true for the technical equipment tools you can use to determine the strengths and weaknesses of each approach.
- A word on replication: It must be noted that writing down the methods section for your research paper must be precise and accurate. Other researchers should be able to replicate your method and come up with the same findings and outcomes. Replication is critical in the research process, as your results must be valid!

STEP 9. Addressing a research paper’s results, analysis, discussion, and conclusion sections
Once you finish the data collection and methods section for your research assignment, it’s high time to move to the results part of your paper and do analysis work. You must use this space as a researcher when dealing with results, analysis, discussion, and conclusion.
The main purpose is to interpret your findings and explain what you could achieve. You can compare your research paper results to prior findings and discuss potential limitations and implications for other researchers.
- Results . Start this section by discussing the findings you could obtain from your research work. Address what research paper questions have been addressed. If you created a series of hypothesis statements or made certain educated guesses in your research paper, state if they have been supported or rejected. It will help if you include tables, graphs, statistical data, and other visual elements to help your readers understand the results.
- Analysis. As a researcher and analyst, you must interpret and explain your findings based on the thesis statement. You have to state whether your research is significant and showcase if your findings support (or reject) prior research findings. Remember to provide a piece of evidence for every fact that you have obtained. Talking in context, explain by comparing and contrasting the facts. It’s also necessary to explain how your findings hold up against other findings conducted by researchers in your field.
- Discussion . Once done with the analytical part, this section should outline and discuss your findings in an accessible way. Remember to mention your research work’s main benefits and limitations. Focus on transparency and allow your readers to determine the weaknesses and specifics of your research. Speaking of tips for writing a research paper analysis part, try to be self-critical but do not overdo it! This part must not introduce any new ideas as you write but help future researchers come up with new methods and try out other things to address the shortcomings of your study.
- Conclusion . This section allows you to summarize and state your thesis again, with a brief overview of the research findings. A conclusion should discuss the main points, starting with the main research questions, the list of methods you have used, the results, and your findings. A research paper’s conclusion should be sufficient to understand the type of work done. Imagine that a reader has no time to read the entire paper and include all the important bits in your conclusion. The key is to write and help everyone understand your content well by reading the conclusion alone!
We are finally ready to write it all down, which will be easier since we already have all the necessary information. As you write a paper for college, the most important thing is to keep the flow and remember to look into your outline. Your research paper must be structured per every section mentioned in your grading rubric and the outline (if you have one!).
Consider starting with an introduction and thinking about starting with the background information bit to present your thesis statement. As you proceed with your writing, a thesis statement should be the last sentence of the introduction part that will help to proceed with the further sections.
Remember the importance of having an outline? If you have been listening and creating one, supporting your thesis with relevant arguments and organizing your content from strong to weak will be easier. Just follow your writing with the sections already mentioned in your outline! Using topic sentences at the beginning that instantly explain your research and include bullet point structures to guide your readers will be helpful.
It also helps to narrow things down and outline the most important ideas and/or facts. A good paper outline will also help you work in chunks and save time, as you only have to grasp some research work simultaneously. It is recommended to allocate at least 2-3 hours to write a research paper to ensure you do not exhaust yourself by spending more as you deal with a lengthy research paper!
A good research paper can be finished, so taking one step at a time for each section will help you see what is left and what is already done. Do your best to avoid procrastination! Meanwhile, as you create a research paper, do not try to reach perfection, as it’s barely possible! Keep yourself disciplined and have a writing schedule, as it will help you to overcome the usual writer’s block issue. It is the best way to meet your deadline and stay on topic! See below for the essential 9 steps that are worth knowing:
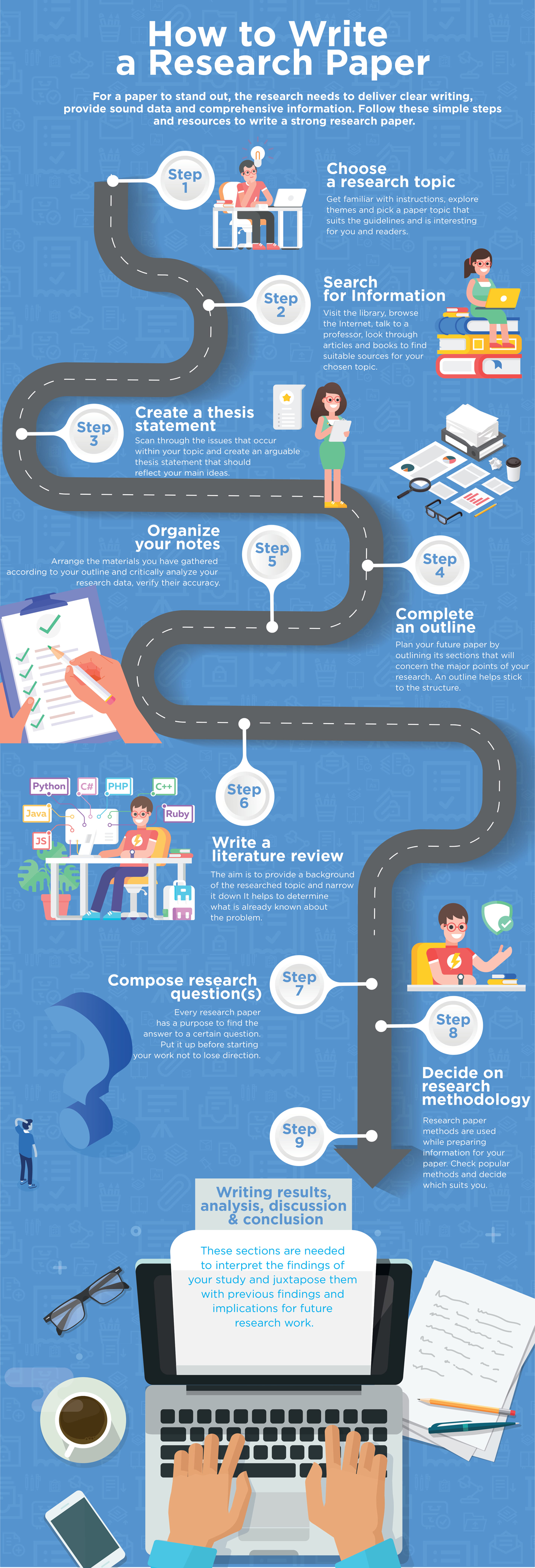
STEP 11. The importance of writing your first draft
It must start with the first aspect of your research paper outline. It means that background information and the relevance of your topic must come first. Make sure to include all your notes and avoid citations unless it’s statistical information that you need to explain the importance of your research. Try to summarize the available information and alternate between paraphrasing and direct quotes as you write a research paper to decrease plagiarism risks.
An ideal scenario would be introducing an issue and continuing with the direct quote, followed by your analysis. Alternatively, writing a science research paper can use various techniques that fit you, like summaries, quotes, tables, or comparisons. Since it’s your first draft, do not spend too much time editing things yet!
Once you are ready with the first draft, remember that it does not have to be perfect because it is not meant for submission. As a rule, most students go through at least three different revisions until they submit the final paper version. Always follow your thesis statement to guarantee that you do not touch upon other subjects.
If you have an opportunity, ask a good friend or even a professional researcher to listen to your draft and help you identify the weaknesses or entire passages to change and improve. It has to be accessible and easy to understand. Always give your first draft enough time, and remember to take a break from your paper and get outside before you get back to it and continue with the revision.
STEP 12. Editing your paper
We are finally at the stage where it is necessary to review the research paper draft and ensure that everything is correct, adheres to the writing standards, and follows existing instructions and/or grading rubrics. To simplify things for any research project, we have created several checklists for you. These include helpful tips and tricks for writing research papers well. They allow you to save time and impress even the most demanding professors.
The main purpose of reviewing your work is to check your paper for any content and logical mistakes. Double-check all the facts and figures. Check your outline and arrange or rearrange ideas based on your notes. You must keep things logical and clear as you edit your paper for repetitions but remember to include all the necessary bibliography and additional notes when and if necessary.
First checklist for writing an excellent paper:
- Is my thesis statement clear and concise for the audience?
- Is the outline followed? Has anything been missed?
- Are arguments presented from strong to weak in a logical way?
- Are all in-text citations corresponding with the Bibliography page?
- Is the thesis supported with strong statements?
- Are intentions and methodology parts clear as to research objectives?
Note: Remember the importance of editing your paper for grammar and spelling mistakes. You can use a dictionary to check spelling and consult a thesaurus if necessary. MS Word and Google Docs allow you to check your spelling for typos and punctuation issues. Take your time to correct the mistakes and proofread your content aloud to ensure it is accessible. If possible, have another person proofread and check your paper, as you may have missed certain weak points. It’s only natural as you write a research paper and edit it repeatedly!
Second checklist for writing:
- Does every body’s paragraph start with a relevant topic sentence?
- Are my arguments supported with evidence and practical examples?
- Have run-on or odd sentences been eliminated?
- Have repetition issues been removed?
- Is the length of sentences normal?
- Is there an easy flow of ideas?
- Has the content been checked for grammar and spelling?
- Are citations accurate and in the correct citation style?
- Is my research paper unbiased and objective?
- Does the paper provide a strong sense of completion?
Note: It is recommended to use “cannot” instead of “can’t” as you write a research paper, as well as “do not” instead of “don’t”. A research paper must be written in the third person unless specified otherwise.
Keeping your writing in style
If you wish to improve your English composition skills, check out “The Elements of Style” by William Strunk for an example of a classic book that addresses style issues in writing. The book’s contents focus on the main grammar rules, elementary principles of composition, useful words and expressions that are usually misused, and important reminders. The book teaches how to revise and rewrite the odd parts. It helps to learn how to avoid all the fancy words. See details of the book by checking The Elements of Style by William Strunk, Jr. , which is partially available online free of charge.
Correct citation and formatting issues
As you might already know, every research paper is written in an academic style like APA , MLA , Chicago , or Harvard. Students majoring in Healthcare will likely use AMA (American Medical Association) citation style. It will depend on your discipline or the style specified in your grading rubric. The most common styles are APA and MLA. The research paper styles that are uncommon are Harvard, Chicago Manual of Style, APSA (American Political Science Association), and the IEEE, which stands for the Institute of Electrical and Electronics Engineers. You can check style guides before you check your formatting aspect.
- APA (American Psychological Association) style is mostly used for social sciences subjects. Make sure to check your grading rubric and university templates for more information.
- MLA (Modern Language Association) style is the most common for Liberal Arts and Humanities. The most recent is MLA 8th Handbook style. Remember to check information on parenthetical citations in MLA to ensure that your research paper follows all the rules. If you are new to this style, check relevant examples for printed books, online sources, etc.
STEP 13. Tools for research paper writing help and accuracy
Once you are done with the editing part and review your draft more than once, it’s time to apply another evaluation method, also known as copy-editing. Thanks to AI-based tools, copy editing is now a more expensive and demanding task. Of course, it will work differently than professional editing because you should still check all the false corrections. Still, since it’s possible free of charge, there is little to complain about when you can write a research paper correctly. You can easily find These types of tools online:
- Grammar checkers. They mostly correct issues like grammar, spelling, typos, and punctuation. It will also address lexical issues, a saving grace for most ESL learners. Remember that poor grammar may seriously ruin your paper, so check grammar checkers and enable them in Google Docs and MS Word.
- Plagiarism checkers . It provides excellent help because you can scan your paper for similarities and balance direct citations and paraphrasing. It does not take more than five minutes to check things, so do yourself a favor and check one of the free or commercial ones. As a researcher, remember to cite your paper completely and give credit where it’s due.
- Citation generators. These are helpful for cases when you cannot find ready-made citation information. When you compose your Works Cited or Bibliography pages, you must double-check the accuracy of citations with all the spaces, punctuation, and indents. The most common research paper styles such tools support are APA, Harvard citations, MLA, Chicago, Vancouver, and more. Free research paper tools also make it possible to convert your sources from one style to another automatically. It is helpful when you have found a complete citation in Chicago but need it in MLA or Vancouver.
- Title page generators. These are useful when brainstorming ideas for a good and inspiring title. After all, a title is what your professor will see first as you submit your paper. Many college students feel challenged as they work with all the indents and ideas, title case problems, and other formatting issues. Title generators help with this problem and will follow the relevant style.
Note: if you plan to get your research paper ready for publishing, read the eligibility rules twice and submit it for peer review. It is also necessary when you plan to attend a scientific conference. As a researcher, you must read and follow all the editorial guidelines before you submit a publication. If you fail to follow the guidelines, you may get rejected even if your paper is an excellent example of academic research.
STEP 14. Final words of encouragement
As we have reached the end of our guide to writing a research paper in 14 easy steps, we sincerely hope that you do not see it as a daunting and frustrating task any longer. Follow every section of our guide and take one step at a time to make things easier. The most important is choosing an inspiring topic you know well, as it’s already half of the task done. Take your time and research the subject that motivates you the most, as it will help you develop a message that makes a difference.
Once again, always pay attention to the literature review part and online research! It will help you to learn what subjects are popular and which need more effort and additional research. It always helps to narrow things down and find something you know well to occupy a research niche.
If you want to contribute something special, consider choosing something not widely researched. Once you are set on a topic for your research paper, think of a strong thesis statement and create an outline. It will help you see the entire picture and clearly understand what steps to take and in what order. It will eventually lead you to an excellent research paper!
If you face any challenges or feel lost when writing a research paper, our trained writers and editors are always ready to help you 24/7!

- Writing a Research Paper
- Research Paper Title
- Research Paper Sources
- Research Paper Problem Statement
- Research Paper Thesis Statement
- Hypothesis for a Research Paper
- Research Question
- Research Paper Outline
- Research Paper Summary
- Research Paper Prospectus
- Research Paper Proposal
- Research Paper Format
- Research Paper Styles
- AMA Style Research Paper
- MLA Style Research Paper
- Chicago Style Research Paper
- APA Style Research Paper
- Research Paper Structure
- Research Paper Cover Page
- Research Paper Abstract
- Research Paper Introduction
- Research Paper Body Paragraph
- Research Paper Literature Review
- Research Paper Background
- Research Paper Methods Section
- Research Paper Results Section
- Research Paper Discussion Section
- Research Paper Conclusion
- Research Paper Appendix
- Research Paper Bibliography
- APA Reference Page
- Annotated Bibliography
- Bibliography vs Works Cited vs References Page
- Research Paper Types
- What is Qualitative Research
Receive paper in 3 Hours!
- Choose the number of pages.
- Select your deadline.
- Complete your order.
Number of Pages
550 words (double spaced)
Deadline: 10 days left
By clicking "Log In", you agree to our terms of service and privacy policy . We'll occasionally send you account related and promo emails.
Sign Up for your FREE account
Loading metrics
Open Access
Ten Simple Rules for Writing Research Papers
* E-mail: [email protected]
Affiliation Department of Computer Science and Engineering, Department of Genetics, Washington University in St. Louis, St. Louis, Missouri, United States of America
- Weixiong Zhang

Published: January 30, 2014
- https://doi.org/10.1371/journal.pcbi.1003453
- Reader Comments
Citation: Zhang W (2014) Ten Simple Rules for Writing Research Papers. PLoS Comput Biol 10(1): e1003453. https://doi.org/10.1371/journal.pcbi.1003453
Editor: Philip E. Bourne, University of California San Diego, United States of America
Copyright: © 2014 Weixiong Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License ., which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Funding: The author received no specific funding for this article.
Competing interests: The author has declared that no competing interests exist.
The importance of writing well can never be overstated for a successful professional career, and the ability to write solid papers is an essential trait of a productive researcher. Writing and publishing a paper has its own life cycle; properly following a course of action and avoiding missteps can be vital to the overall success not only of a paper but of the underlying research as well. Here, we offer ten simple rules for writing and publishing research papers.
As a caveat, this essay is not about the mechanics of composing a paper, much of which has been covered elsewhere, e.g., [1] , [2] . Rather, it is about the principles and attitude that can help guide the process of writing in particular and research in general. In this regard, some of the discussion will complement, extend, and refine some advice given in early articles of this Ten Simple Rules series of PLOS Computational Biology [3] – [8] .
Rule 1: Make It a Driving Force
Never separate writing a paper from the underlying research. After all, writing and research are integral parts of the overall enterprise. Therefore, design a project with an ultimate paper firmly in mind. Include an outline of the paper in the initial project design documents to help form the research objectives, determine the logical flow of the experiments, and organize the materials and data to be used. Furthermore, use writing as a tool to reassess the overall project, reevaluate the logic of the experiments, and examine the validity of the results during the research. As a result, the overall research may need to be adjusted, the project design may be revised, new methods may be devised, and new data may be collected. The process of research and writing may be repeated if necessary.
Rule 2: Less Is More
It is often the case that more than one hypothesis or objective may be tackled in one project. It is also not uncommon that the data and results gathered for one objective can serve additional purposes. A decision on having one or more papers needs to be made, and the decision will be affected by various factors. Regardless of the validity of these factors, the overriding consideration must be the potential impact that the paper may have on the research subject and field. Therefore, the significance, completeness, and coherence of the results presented as a whole should be the principal guide for selecting the story to tell, the hypothesis to focus upon, and materials to include in the paper, as well as the yardstick for measuring the quality of the paper. By this metric, less is more , i.e., fewer but more significant papers serve both the research community and one's career better than more papers of less significance.
Rule 3: Pick the Right Audience
Deciding on an angle of the story to focus upon is the next hurdle to jump at the initial stage of the writing. The results from a computational study of a biological problem can often be presented to biologists, computational scientists, or both; deciding what story to tell and from what angle to pitch the main idea is important. This issue translates to choosing a target audience, as well as an appropriate journal, to cast the main messages to. This is critical for determining the organization of the paper and the level of detail of the story, so as to write the paper with the audience in mind. Indeed, writing a paper for biologists in general is different from writing for specialists in computational biology.
Rule 4: Be Logical
The foundation of “lively” writing for smooth reading is a sound and clear logic underlying the story of the paper. Although experiments may be carried out independently, the result from one experiment may form premises and/or provide supporting data for the next experiment. The experiments and results, therefore, must be presented in a logical order. In order to make the writing an easy process to follow, this logical flow should be determined before any other writing strategy or tactic is exercised. This logical order can also help you avoid discussing the same issue or presenting the same argument in multiple places in the paper, which may dilute the readers' attention.
An effective tactic to help develop a sound logical flow is to imaginatively create a set of figures and tables, which will ultimately be developed from experimental results, and order them in a logical way based on the information flow through the experiments. In other words, the figures and tables alone can tell the story without consulting additional material. If all or some of these figures and tables are included in the final manuscript, make every effort to make them self-contained (see Rule 5 below), a favorable feature for the paper to have. In addition, these figures and tables, as well as the threading logical flow, may be used to direct or organize research activities, reinforcing Rule 1.
Rule 5: Be Thorough and Make It Complete
Completeness is a cornerstone for a research paper, following Rule 2. This cornerstone needs to be set in both content and presentation. First, important and relevant aspects of a hypothesis pursued in the research should be discussed with detailed supporting data. If the page limit is an issue, focus on one or two main aspects with sufficient details in the main text and leave the rest to online supporting materials. As a reminder, be sure to keep the details of all experiments (e.g., parameters of the experiments and versions of software) for revision, post-publication correspondence, or importantly, reproducibility of the results. Second, don't simply state what results are presented in figures and tables, which makes the writing repetitive because they are self-contained (see below), but rather, interpret them with insights to the underlying story to be told (typically in the results section) and discuss their implication (typically in the discussion section).
Third, make the whole paper self-contained. Introduce an adequate amount of background and introductory material for the right audience (following Rule 3). A statistical test, e.g., hypergeometric tests for enrichment of a subset of objects, may be obvious to statisticians or computational biologists but may be foreign to others, so providing a sufficient amount of background is the key for delivery of the material. When an uncommon term is used, give a definition besides a reference to it. Fourth, try to avoid “making your readers do the arithmetic” [9] , i.e., be clear enough so that the readers don't have to make any inference from the presented data. If such results need to be discussed, make them explicit even though they may be readily derived from other data. Fifth, figures and tables are essential components of a paper, each of which must be included for a good reason; make each of them self-contained with all required information clearly specified in the legend to guide interpretation of the data presented.
Rule 6: Be Concise
This is a caveat to Rule 5 and is singled out to emphasize its importance. Being thorough is not a license to writing that is unnecessarily descriptive, repetitive, or lengthy. Rather, on the contrary, “simplicity is the ultimate sophistication” [10] . Overly elaborate writing is distracting and boring and places a burden on the readers. In contrast, the delivery of a message is more rigorous if the writing is precise and concise. One excellent example is Watson and Crick's Nobel-Prize-winning paper on the DNA double helix structure [11] —it is only two pages long!
Rule 7: Be Artistic
A complete draft of a paper requires a lot of work, so it pays to go the extra mile to polish it to facilitate enjoyable reading. A paper presented as a piece of art will give referees a positive initial impression of your passion toward the research and the quality of the work, which will work in your favor in the reviewing process. Therefore, concentrate on spelling, grammar, usage, and a “lively” writing style that avoids successions of simple, boring, declarative sentences. Have an authoritative dictionary with a thesaurus and a style manual, e.g., [1] , handy and use them relentlessly. Also pay attention to small details in presentation, such as paragraph indentation, page margins, and fonts. If you are not a native speaker of the language the paper is written in, make sure to have a native speaker go over the final draft to ensure correctness and accuracy of the language used.
Rule 8: Be Your Own Judge
A complete manuscript typically requires many rounds of revision. Taking a correct attitude during revision is critical to the resolution of most problems in the writing. Be objective and honest about your work and do not exaggerate or belittle the significance of the results and the elegance of the methods developed. After working long and hard, you are an expert on the problem you studied, and you are the best referee of your own work, after all . Therefore, inspect the research and the paper in the context of the state of the art.
When revising a draft, purge yourself out of the picture and leave your passion for your work aside. To be concrete, put yourself completely in the shoes of a referee and scrutinize all the pieces—the significance of the work, the logic of the story, the correctness of the results and conclusions, the organization of the paper, and the presentation of the materials. In practice, you may put a draft aside for a day or two—try to forget about it completely—and then come back to it fresh, consider it as if it were someone else's writing, and read it through while trying to poke holes in the story and writing. In this process, extract the meaning literally from the language as written and do not try to use your own view to interpret or extrapolate from what was written. Don't be afraid to throw away pieces of your writing and start over from scratch if they do not pass this “not-yourself” test. This can be painful, but the final manuscript will be more logically sound and better organized.
Rule 9: Test the Water in Your Own Backyard
It is wise to anticipate the possible questions and critiques the referees may raise and preemptively address their concerns before submission. To do so, collect feedback and critiques from others, e.g., colleagues and collaborators. Discuss your work with them and get their opinions, suggestions, and comments. A talk at a lab meeting or a departmental seminar will also help rectify potential issues that need to be addressed. If you are a graduate student, running the paper and results through the thesis committee may be effective to iron out possible problems.
Rule 10: Build a Virtual Team of Collaborators
When a submission is rejected or poorly reviewed, don't be offended and don't take it personally. Be aware that the referees spent their time on the paper, which they might have otherwise devoted to their own research, so they are doing you a favor and helping you shape the paper to be more accessible to the targeted audience. Therefore, consider the referees as your collaborators and treat the reviews with respect. This attitude can improve the quality of your paper and research.
Read and examine the reviews objectively—the principles set in Rule 8 apply here as well. Often a criticism was raised because one of the aspects of a hypothesis was not adequately studied, or an important result from previous research was not mentioned or not consistent with yours. If a critique is about the robustness of a method used or the validity of a result, often the research needs to be redone or more data need to be collected. If you believe the referee has misunderstood a particular point, check the writing. It is often the case that improper wording or presentation misled the referee. If that's the case, revise the writing thoroughly. Don't argue without supporting data. Don't submit the paper elsewhere without additional work. This can only temporally mitigate the issue, you will not be happy with the paper in the long run, and this may hurt your reputation.
Finally, keep in mind that writing is personal, and it takes a lot of practice to find one's style. What works and what does not work vary from person to person. Undoubtedly, dedicated practice will help produce stronger papers with long-lasting impact.
Acknowledgments
Thanks to Sharlee Climer, Richard Korf, and Kevin Zhang for critical reading of the manuscript.
- 1. Strunk W Jr, White EB (1999) The Elements of Style. 4th edition. New York: Longman.
- 2. Zinsser W (2006) On Writing Well: The Classic Guide to Writing Nonfiction. 30th anniversary edition. New York: Harper Perennial.
- View Article
- Google Scholar
- 9. Johnson DS (2002) A theoretician's guide to the experimental analysis of algorithms. In Goldwasser MH, Johnson DS, McGeoch CC, editors. Data Structures, Near Neighbor Searches, and Methodology: Fifth and Sixth DIMACS Implementation Challenges. Providence: American Mathematical Society. pp.215–250.
- 10. Wikiquote page on Leonardo Da Vinci. Available: http://en.wikiquote.org/wiki/Leonardo_da_Vinci#Quotes_about_Leonardo . Accessed 13 December 2013.
- Thesis Action Plan New
- Academic Project Planner
Literature Navigator
Thesis dialogue blueprint, writing wizard's template, research proposal compass.
- Why students love us
- Rebels Blog
- Why we are different
- All Products
- Coming Soon
How to Start Research Paper: A Comprehensive Guide for Students

Writing a research paper can feel like a big challenge, but it doesn't have to be! This guide will help you understand the steps needed to create a great research paper. From picking a topic to writing your introduction, we’ll cover everything you need to know to get started on the right foot.
Key Takeaways
- Understanding the structure of a research paper is essential; each section serves a specific purpose.
- Choosing a topic that interests you will keep you motivated throughout your research.
- Doing preliminary research helps you find credible sources and key literature.
- A strong thesis statement clearly defines your research question and argument.
- Properly citing sources is crucial to avoid plagiarism and maintain academic integrity.
Understanding The Research Paper Structure

A research paper is a carefully organized document that presents the results of your investigation on a specific topic. Understanding its structure is crucial for effective writing. This structure typically includes several key components:
Components Of A Research Paper
- Title Page : This is the first page, containing the title of your paper, your name, and other relevant details.
- Abstract : A brief summary of your research, usually around 150-250 words.
- Introduction : This section introduces your topic, provides background information, and states your thesis.
- Literature Review : Here, you summarize existing research related to your topic, highlighting gaps your study will address.
- Methodology : This part explains how you conducted your research, including data collection and analysis methods.
- Results : Present the findings of your research, often with tables or graphs for clarity.
- Discussion : Analyze your results, discussing their implications and how they relate to your thesis.
- Conclusion : Summarize your findings and suggest areas for future research.
- References : A list of all the sources you cited in your paper.
Importance Of Each Section
Each section of a research paper serves a specific purpose:
- The introduction sets the stage for your research.
- The literature review shows your understanding of the field.
- The methodology provides transparency about your research process.
- The results and discussion sections allow you to present and interpret your findings.
- The conclusion ties everything together and emphasizes the significance of your work.
Common Formatting Styles
Different academic fields often require specific formatting styles. Here are a few common ones:
- APA (American Psychological Association) : Common in social sciences.
- MLA (Modern Language Association) : Often used in humanities.
- Chicago/Turabian : Used in history and some other disciplines.
Understanding these components and their importance will help you create a well-structured research paper. For additional support, consider resources like the Research Proposal Compass , which offers templates and guidance for crafting high-quality proposals. This can ease the process and enhance your academic success.
Selecting An Engaging Research Topic
Choosing a research topic is a crucial step in your academic journey. This decision shapes the entire direction of your paper. Here are some key aspects to consider:
Identifying Your Interests
Start by reflecting on what excites you. Make a list of subjects that you find enjoyable and fulfilling. This will help you stay motivated throughout your research. Think about how these interests can connect to your field of study.
Evaluating Topic Feasibility
Once you have a list of potential topics, assess their feasibility. Consider the availability of data and resources. A realistic topic will make it easier to support your arguments and structure your paper effectively. For example, if you’re interested in childhood diseases, narrow it down to something specific like juvenile diabetes. This will help you focus your research and make it more manageable.
Narrowing Down Your Focus
To refine your topic, break it down into smaller parts. Here’s a quick checklist to help you narrow it down:
- Identify the core aspects of your subject.
- Look for subtopics or issues within the main topic.
- Consider different perspectives or angles.
- Determine the scope of your research.
By aligning your topic with your interests and ensuring it has enough resources, you set the stage for a research paper that is both engaging and academically sound. Don't hesitate to consult resources like the Thesis Action Plan by Research Rebels for structured guidance on topic selection and research strategies. Remember, a well-chosen topic is the foundation of a successful research paper!
Conducting Preliminary Research
Before you start writing your research paper, it's essential to conduct preliminary research. This step helps you gather a broad understanding of your topic and ensures you have a solid foundation for your work. Starting early can save you time and effort later on.
Utilizing Academic Databases
Begin by exploring various academic databases and library resources. These platforms provide access to a wealth of scholarly articles, books, and other materials. Here are some steps to guide you:
- Identify relevant databases for your field of study.
- Use specific keywords related to your topic to narrow down your search.
- Skim through the results to find useful sources, setting aside those that seem promising for a deeper read later.
Identifying Key Literature
As you gather information, focus on identifying key literature that relates to your topic. This includes:
- Primary sources : Firsthand accounts like original research articles.
- Secondary sources : Reviews or analyses of primary research.
Understanding the difference between these sources is crucial. For instance, the experimental research roadmap can guide you through the complexities of your topic, helping you identify what has already been studied and what gaps exist.
Assessing Source Credibility
Not all sources are created equal. To ensure the quality of your research, evaluate the credibility of each source. Consider the following:
- Authority : Who is the author? Are they an expert in the field?
- Objectivity : Is the source biased, or does it present balanced information?
- Relevance : Does the source directly relate to your research question?
By following these steps, you will lay a solid foundation for your research and be well-prepared to delve into more specific inquiries. Remember, conducting thorough preliminary research is key to developing a focused and effective research paper.
Crafting A Strong Thesis Statement
A strong thesis statement is essential for guiding your research paper. It acts as the backbone of your argument, providing clarity and direction. Your thesis should be specific, debatable, and supported by evidence. Here’s how to create an effective thesis statement:
Defining Your Research Question
- Start by identifying the main question your research addresses. This question should be clear and focused, allowing you to explore it thoroughly.
- Ensure that your question is debatable, meaning others can have different opinions on it.
Articulating Your Argument
- Your thesis statement should clearly express your position on the topic. Avoid vague language and be assertive in your claims.
- For example, instead of saying, "Many people think that climate change is a problem," you could say, "Climate change poses a significant threat to global ecosystems and requires immediate action."
Aligning With Research Objectives
- Make sure your thesis aligns with the objectives of your research. This will help you stay focused and organized as you write.
- Regularly revisit your thesis statement to ensure it still reflects your argument as your paper develops.
In summary, crafting a strong thesis statement is crucial for your research paper. It not only guides your writing but also engages your readers by presenting a clear argument. For more tips on how to write a thesis fast and how to write thesis easily , consider resources like the Thesis Dialogue Blueprint that offer structured approaches to thesis writing. Remember, a well-defined thesis can make a significant difference in the quality of your work!
Developing A Comprehensive Outline
Creating a detailed outline is essential for organizing your research paper effectively. A well-structured outline serves as a roadmap that guides you through the writing process, ensuring that you cover all necessary points and maintain a logical flow.
Structuring Your Ideas
Begin by identifying the main topics and subtopics you want to address in your paper. This can be done through brainstorming or free writing. Here’s a simple approach to get started:
- List your main ideas.
- Break down each idea into subtopics.
- Organize these subtopics in a logical order.
Creating Logical Flow
Once you have your main topics and subtopics, arrange them in a way that makes sense. Consider the following:
- Start with your thesis statement at the top.
- Use Roman numerals (I, II, III) for major sections.
- Label supporting points with capital letters (A, B, C).
- Include any relevant evidence or examples under each point.
This method not only helps in organizing your thoughts but also ensures that you don’t miss any critical information.
Incorporating Supporting Evidence
Don’t forget to include supporting evidence in your outline. This is crucial for backing up your arguments. Here’s how to do it:
- Under each subtopic, jot down key points from your research.
- Note down the sources you plan to use for each point.
- This will help you stay organized and prevent important details from slipping through the cracks.
By following these steps, you can create a comprehensive outline that will make the writing process smoother and more efficient. Remember, a solid outline is the foundation of a successful research paper!
Choosing Appropriate Research Methodologies
When embarking on your research journey, selecting the right methodology is crucial. This choice will shape how you collect and analyze data, ultimately influencing your findings. Your methodology should align with your research question and objectives.
Qualitative vs. Quantitative Approaches
Understanding the difference between qualitative and quantitative methods is essential:
- Qualitative Methods : These focus on exploring ideas and understanding experiences. They often involve interviews, observations, and case studies.
- Quantitative Methods : These are about numbers and statistics. They help test hypotheses and analyze data through surveys and experiments.
- Mixed Methods : Combining both approaches can provide a comprehensive view of your research question.
Selecting Data Collection Methods
Choosing how to gather your data is another key step. Here are some common methods:
| Method | Use Case |
|---|---|
| Survey | Gathering large amounts of standardized data |
| Interview | In-depth understanding of individual perspectives |
| Observation | Real-time insights into behavior |
| Experiment | Establishing cause-and-effect relationships |
Justifying Your Methodology Choice
It's important to explain why you chose a particular methodology. Consider the following:
- How does it fit your research question?
- What are the strengths and weaknesses of your chosen method?
- How will it help you achieve your research objectives?
By carefully selecting and justifying your methodology, you ensure that your research is both credible and impactful. For more guidance, resources like Research Rebels can provide valuable insights into effective research practices.
Writing The Introduction Effectively
Engaging your audience.
To start your introduction, hook your reader with an interesting fact or a thought-provoking question. This initial engagement is crucial as it sets the tone for your entire research paper. Think of it as the trailer for a movie; it should entice your audience to want to know more.
Presenting Background Information
Next, provide some context for your topic. This means explaining why your research is important and what background information your readers need to understand your work. You want to strike a balance—too little context can leave your audience confused, while too much can bore them. Aim for clarity and relevance.
Stating Your Thesis Clearly
Finally, clearly state your thesis statement. This is the core argument of your paper, and it should be concise and direct. Your thesis will guide the rest of your research, so make sure it reflects the main points you will discuss. Remember, a strong thesis is essential for a successful paper.
Quick Checklist for Your Introduction
- Hook your reader with an engaging opening.
- Provide necessary background information .
- Clearly state your thesis.
- Outline your approach to the topic.
By following these steps, you can craft an introduction that not only captures attention but also lays a solid foundation for your research paper. If you're looking for more guidance, consider using resources like the [ Writing Wizard's Template ](https://www.researchrebels.com/products/writing-wizards-template) to help structure your introduction effectively. This can alleviate some of the stress associated with writing your thesis and help you learn how to write dissertation fast .
Conducting A Thorough Literature Review
A literature review is a crucial step in your research journey. It helps you understand what has already been studied and where your work can fit in. By conducting a thorough literature review, you can identify gaps in existing research and build a strong foundation for your own study.
Identifying Gaps In Existing Research
To start, you need to define your research question. This will guide your search for relevant literature. Here are some steps to help you:
- Define your research question clearly.
- Search for current, relevant, and reliable sources using academic databases.
- Analyze the literature critically to find key themes and debates.
- Synthesize findings to identify research gaps that your work could fill.
Synthesizing Key Findings
As you gather information, it’s important to organize your findings. You can use a literature review matrix to help you keep track of key points and themes. This will make it easier to see how different studies relate to each other and to your research question.
| Source | Key Findings | Gaps Identified |
|---|---|---|
| Source 1 | Finding A | Gap 1 |
| Source 2 | Finding B | Gap 2 |
| Source 3 | Finding C | Gap 3 |
Establishing Context For Your Study
Finally, your literature review should not only summarize existing research but also set the stage for your own study. Make sure to connect your findings back to your research question and explain how your work will contribute to the field. This will demonstrate your understanding and help you build credibility.
In summary, a well-conducted literature review is essential for your research paper. It not only informs your study but also shows your engagement with the academic community. For more guidance, consider using tools like the Literature Navigator , which can help streamline your research process and enhance your efficiency.
Analyzing And Interpreting Data
Once you have gathered your data, the next step is to analyze and interpret it. This process involves organizing the data, identifying patterns, and drawing meaningful conclusions. Here’s how you can approach this crucial phase:
Data Analysis Techniques
- Organize your data systematically. This could involve sorting it into categories or using software tools to help manage it.
- Identify patterns and trends. Look for recurring themes or significant changes in your data that can help tell your research story.
- Use statistical analysis for quantitative data. Techniques like t-tests or chi-square tests can help you understand relationships between variables.
- Apply thematic analysis for qualitative data. This method allows you to sift through text and find recurring themes that support your research.
Drawing Conclusions
- Interpret the results in context. Consider how your findings relate to your research questions and the existing literature. For example, if you find that a particular skill is highly sought after in job postings, it might indicate its importance in the job market.
- Ensure your conclusions are valid and reliable. This means double-checking your data and analysis methods to confirm that your findings are accurate.
Presenting Your Findings
To effectively communicate your results, consider using tables and charts. Here’s a simple table format you can use:
| Variable | Description | Results |
|---|---|---|
| Var1 | Description1 | Result1 |
| Var2 | Description2 | Result2 |
Remember, the goal is not just to analyze but to interpret. What do the numbers mean in the context of your research? How do they support your thesis? Resources like "Unlocking the Secrets of Data" can provide further insights into effective data interpretation. Additionally, consider reading "An Engaging Journey Through Statistics" for a more enjoyable approach to understanding data analysis.
Citing Sources And Avoiding Plagiarism
Understanding citation styles.
Citing your sources is essential in academic writing. It not only gives credit to the original authors but also helps you avoid plagiarism. Different citation styles, like APA, MLA, and Chicago, have specific rules for formatting citations. Here’s a quick overview of common styles:
| Style | Description |
|---|---|
| APA | Used mainly in social sciences. |
| MLA | Common in humanities. |
| Chicago | Often used in history and some social sciences. |
Properly Quoting And Paraphrasing
When you use someone else's ideas or words, you must cite them. Here are some tips:
- Quotation : Use the exact words from a source, placing them in quotation marks and citing the source.
- Paraphrasing : Rewrite the idea in your own words while still giving credit to the original author.
- Summarizing : Condense the main ideas of a source into a brief overview, and cite it.
Maintaining Academic Integrity
To keep your work original and ethical:
- Understand what plagiarism is and how to avoid it.
- Use plagiarism detection tools to check your work before submission.
- Always cite your sources, even if you’re unsure. It’s better to over-cite than to risk plagiarism.
By following these guidelines, you can ensure that your research paper is both credible and respectful of others' work. Remember, using tools like WhatsApp to discuss your findings with peers can also help clarify your understanding of proper citation practices.
In summary, proper citation is not just a formality; it’s a crucial part of academic writing that upholds integrity and respect for intellectual property.
Revising And Editing Your Draft

Revising and editing your draft is a crucial step in the writing process. This is where you refine your ideas and ensure your arguments are clear and compelling. Taking the time to revise can significantly enhance the quality of your research paper.
Seeking Feedback From Peers
- Share your draft with classmates or friends. Their fresh perspectives can help you identify areas that need improvement.
- Consider joining a study group where you can exchange feedback on each other's work.
Improving Clarity And Coherence
- Read your paper out loud. This can help you catch awkward phrases and unclear sentences.
- Check if each paragraph supports your thesis statement. If a paragraph doesn’t relate, consider revising or removing it.
Finalizing Your Research Paper
- After making revisions, proofread your paper for grammar and spelling errors. Tools like Grammarly can assist in this process.
- Ensure that all your sources are properly cited to maintain academic integrity . This is essential to avoid plagiarism, which can undermine your hard work.
By following these steps, you can transform your draft into a polished final product. For more detailed guidance, consider resources like the Interview Research Roadmap for mastering interview techniques or the Thesis Success Essentials for templates that can help streamline your writing process.
When you're done with your first draft, it's time to make it shine! Revising and editing are key steps that can turn a good paper into a great one. Don't let confusion hold you back— visit our website for helpful tips and tools that can guide you through the process. Start your journey to a polished thesis today!
In conclusion, starting a research paper can feel overwhelming, but it doesn't have to be. By following the steps outlined in this guide, students can approach the task with confidence. From picking a topic that interests you to organizing your research and writing clearly, each part is important. Remember, this process is not just about finishing an assignment; it's about learning and sharing your ideas with others. As you work on your paper, keep in mind that every step you take helps you grow as a writer and thinker. Embrace the challenge, and let your unique voice shine through in your work. Happy writing!
Frequently Asked Questions
What is a research paper.
A research paper is a detailed piece of writing where you explore a topic, gather information, and present your findings. It shows what you've learned about a subject.
How do I choose a good topic for my research paper?
Pick a topic that interests you and has enough information available. Make sure it’s not too broad or too narrow.
What should I include in the introduction of my research paper?
In the introduction, you should introduce your topic, explain why it’s important, and state your main argument or thesis.
How do I create a strong thesis statement?
A strong thesis statement clearly expresses your main point or argument in one or two sentences. It should be specific and guide your research.
What is the purpose of a literature review?
A literature review summarizes existing research on your topic. It helps you understand what has already been studied and where your work fits in.
How do I avoid plagiarism in my research paper?
To avoid plagiarism, always give credit to the original authors when you use their ideas or words. Use proper citations for quotes and paraphrased content.
What are some tips for writing a strong conclusion?
In your conclusion, summarize your main points, restate your thesis in a new way, and discuss the significance of your findings.
How can I improve my research paper before submitting it?
Revise and edit your paper carefully. Check for clarity, grammar, and spelling. Getting feedback from peers or teachers can also help improve your work.

Discovering Statistics Using IBM SPSS Statistics: A Fun and Informative Guide

Unlocking the Power of Data: A Review of 'Essentials of Modern Business Statistics with Microsoft Excel'

Discovering Statistics Using SAS: A Comprehensive Review

How to Research for Your Thesis Paper Without Feeling Overwhelmed

Collaborative Genius: 6 Hacks for Seamless Group Thesis Writing

Understanding the Difference Between Research Objectives and Research Questions

Thesis Action Plan

- Blog Articles
- Affiliate Program
- Terms and Conditions
- Payment and Shipping Terms
- Privacy Policy
- Return Policy
© 2024 Research Rebels, All rights reserved.
Your cart is currently empty.
Ten Simple Rules for Writing Research Papers
- 10(1):e1003453
- This person is not on ResearchGate, or hasn't claimed this research yet.
Discover the world's research
- 25+ million members
- 160+ million publication pages
- 2.3+ billion citations

- PLOS COMPUT BIOL

- David M Evans

- Mike Chalupa
- Abigail M. Newbury
- J TRANSL MED

- Steven E. Brenner
- Michael B Eisen

- J.D. Watson
- William K. Zinsser

- James D. Watson
- Francis H. C. Crick
- William Strunk
- E. B. White
- ANN INTERN MED
- J. D. Watson
- F. H. C. Crick
- Recruit researchers
- Join for free
- Login Email Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google Welcome back! Please log in. Email · Hint Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google No account? Sign up
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
The PMC website is updating on October 15, 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- PLoS Comput Biol
- v.18(6); 2022 Jun

Ten simple rules for good research practice
Simon schwab.
1 Center for Reproducible Science, University of Zurich, Zurich, Switzerland
2 Epidemiology, Biostatistics and Prevention Institute, University of Zurich, Zurich, Switzerland
Perrine Janiaud
3 Department of Clinical Research, University Hospital Basel, University of Basel, Basel, Switzerland
Michael Dayan
4 Human Neuroscience Platform, Fondation Campus Biotech Geneva, Geneva, Switzerland
Valentin Amrhein
5 Department of Environmental Sciences, Zoology, University of Basel, Basel, Switzerland
Radoslaw Panczak
6 Institute of Social and Preventive Medicine, University of Bern, Bern, Switzerland
Patricia M. Palagi
7 SIB Training Group, SIB Swiss Institute of Bioinformatics, Lausanne, Switzerland
Lars G. Hemkens
8 Meta-Research Innovation Center at Stanford (METRICS), Stanford University, Stanford, California, United States of America
9 Meta-Research Innovation Center Berlin (METRIC-B), Berlin Institute of Health, Berlin, Germany
Meike Ramon
10 Applied Face Cognition Lab, University of Lausanne, Lausanne, Switzerland
Nicolas Rothen
11 Faculty of Psychology, UniDistance Suisse, Brig, Switzerland
Stephen Senn
12 Statistical Consultant, Edinburgh, United Kingdom
Leonhard Held
This is a PLOS Computational Biology Methods paper.
Introduction
The lack of research reproducibility has caused growing concern across various scientific fields [ 1 – 5 ]. Today, there is widespread agreement, within and outside academia, that scientific research is suffering from a reproducibility crisis [ 6 , 7 ]. Researchers reach different conclusions—even when the same data have been processed—simply due to varied analytical procedures [ 8 , 9 ]. As we continue to recognize this problematic situation, some major causes of irreproducible research have been identified. This, in turn, provides the foundation for improvement by identifying and advocating for good research practices (GRPs). Indeed, powerful solutions are available, for example, preregistration of study protocols and statistical analysis plans, sharing of data and analysis code, and adherence to reporting guidelines. Although these and other best practices may facilitate reproducible research and increase trust in science, it remains the responsibility of researchers themselves to actively integrate them into their everyday research practices.
Contrary to ubiquitous specialized training, cross-disciplinary courses focusing on best practices to enhance the quality of research are lacking at universities and are urgently needed. The intersections between disciplines offer a space for peer evaluation, mutual learning, and sharing of best practices. In medical research, interdisciplinary work is inevitable. For example, conducting clinical trials requires experts with diverse backgrounds, including clinical medicine, pharmacology, biostatistics, evidence synthesis, nursing, and implementation science. Bringing researchers with diverse backgrounds and levels of experience together to exchange knowledge and learn about problems and solutions adds value and improves the quality of research.
The present selection of rules was based on our experiences with teaching GRP courses at the University of Zurich, our course participants’ feedback, and the views of a cross-disciplinary group of experts from within the Swiss Reproducibility Network ( www.swissrn.org ). The list is neither exhaustive, nor does it aim to address and systematically summarize the wide spectrum of issues including research ethics and legal aspects (e.g., related to misconduct, conflicts of interests, and scientific integrity). Instead, we focused on practical advice at the different stages of everyday research: from planning and execution to reporting of research. For a more comprehensive overview on GRPs, we point to the United Kingdom’s Medical Research Council’s guidelines [ 10 ] and the Swedish Research Council’s report [ 11 ]. While the discussion of the rules may predominantly focus on clinical research, much applies, in principle, to basic biomedical research and research in other domains as well.
The 10 proposed rules can serve multiple purposes: an introduction for researchers to relevant concepts to improve research quality, a primer for early-career researchers who participate in our GRP courses, or a starting point for lecturers who plan a GRP course at their own institutions. The 10 rules are grouped according to planning (5 rules), execution (3 rules), and reporting of research (2 rules); see Fig 1 . These principles can (and should) be implemented as a habit in everyday research, just like toothbrushing.

GRP, good research practices.
Research planning
Rule 1: specify your research question.
Coming up with a research question is not always simple and may take time. A successful study requires a narrow and clear research question. In evidence-based research, prior studies are assessed in a systematic and transparent way to identify a research gap for a new study that answers a question that matters [ 12 ]. Papers that provide a comprehensive overview of the current state of research in the field are particularly helpful—for example, systematic reviews. Perspective papers may also be useful, for example, there is a paper with the title “SARS-CoV-2 and COVID-19: The most important research questions.” However, a systematic assessment of research gaps deserves more attention than opinion-based publications.
In the next step, a vague research question should be further developed and refined. In clinical research and evidence-based medicine, there is an approach called population, intervention, comparator, outcome, and time frame (PICOT) with a set of criteria that can help framing a research question [ 13 ]. From a well-developed research question, subsequent steps will follow, which may include the exact definition of the population, the outcome, the data to be collected, and the sample size that is required. It may be useful to find out if other researchers find the idea interesting as well and whether it might promise a valuable contribution to the field. However, actively involving the public or the patients can be a more effective way to determine what research questions matter.
The level of details in a research question also depends on whether the planned research is confirmatory or exploratory. In contrast to confirmatory research, exploratory research does not require a well-defined hypothesis from the start. Some examples of exploratory experiments are those based on omics and multi-omics experiments (genomics, bulk RNA-Seq, single-cell, etc.) in systems biology and connectomics and whole-brain analyses in brain imaging. Both exploration and confirmation are needed in science, and it is helpful to understand their strengths and limitations [ 14 , 15 ].
Rule 2: Write and register a study protocol
In clinical research, registration of clinical trials has become a standard since the late 1990 and is now a legal requirement in many countries. Such studies require a study protocol to be registered, for example, with ClinicalTrials.gov, the European Clinical Trials Register, or the World Health Organization’s International Clinical Trials Registry Platform. Similar effort has been implemented for registration of systematic reviews (PROSPERO). Study registration has also been proposed for observational studies [ 16 ] and more recently in preclinical animal research [ 17 ] and is now being advocated across disciplines under the term “preregistration” [ 18 , 19 ].
Study protocols typically document at minimum the research question and hypothesis, a description of the population, the targeted sample size, the inclusion/exclusion criteria, the study design, the data collection, the data processing and transformation, and the planned statistical analyses. The registration of study protocols reduces publication bias and hindsight bias and can safeguard honest research and minimize waste of research [ 20 – 22 ]. Registration ensures that studies can be scrutinized by comparing the reported research with what was actually planned and written in the protocol, and any discrepancies may indicate serious problems (e.g., outcome switching).
Note that registration does not mean that researchers have no flexibility to adapt the plan as needed. Indeed, new or more appropriate procedures may become available or known only after registration of a study. Therefore, a more detailed statistical analysis plan can be amended to the protocol before the data are observed or unblinded [ 23 , 24 ]. Likewise, registration does not exclude the possibility to conduct exploratory data analyses; however, they must be clearly reported as such.
To go even further, registered reports are a novel article type that incentivize high-quality research—irrespective of the ultimate study outcome [ 25 , 26 ]. With registered reports, peer-reviewers decide before anyone knows the results of the study, and they have a more active role in being able to influence the design and analysis of the study. Journals from various disciplines increasingly support registered reports [ 27 ].
Naturally, preregistration and registered reports also have their limitations and may not be appropriate in a purely hypothesis-generating (explorative) framework. Reports of exploratory studies should indeed not be molded into a confirmatory framework; appropriate rigorous reporting alternatives have been suggested and start to become implemented [ 28 , 29 ].
Rule 3: Justify your sample size
Early-career researchers in our GRP courses often identify sample size as an issue in their research. For example, they say that they work with a low number of samples due to slow growth of cells, or they have a limited number of patient tumor samples due to a rare disease. But if your sample size is too low, your study has a high risk of providing a false negative result (type II error). In other words, you are unlikely to find an effect even if there truly was an effect.
Unfortunately, there is more bad news with small studies. When an effect from a small study was selected for drawing conclusions because it was statistically significant, low power increases the probability that an effect size is overestimated [ 30 , 31 ]. The reason is that with low power, studies that due to sampling variation find larger (overestimated) effects are much more likely to be statistically significant than those that happen to find smaller (more realistic) effects [ 30 , 32 , 33 ]. Thus, in such situations, effect sizes are often overestimated. For the phenomenon that small studies often report more extreme results (in meta-analyses), the term “small-study effect” was introduced [ 34 ]. In any case, an underpowered study is a problematic study, no matter the outcome.
In conclusion, small sample sizes can undermine research, but when is a study too small? For one study, a total of 50 patients may be fine, but for another, 1,000 patients may be required. How large a study needs to be designed requires an appropriate sample size calculation. Appropriate sample size calculation ensures that enough data are collected to ensure sufficient statistical power (the probability to reject the null hypothesis when it is in fact false).
Low-powered studies can be avoided by performing a sample size calculation to find out the required sample size of the study. This requires specifying a primary outcome variable and the magnitude of effect you are interested in (among some other factors); in clinical research, this is often the minimal clinically relevant difference. The statistical power is often set at 80% or larger. A comprehensive list of packages for sample size calculation are available [ 35 ], among them the R package “pwr” [ 36 ]. There are also many online calculators available, for example, the University of Zurich’s “SampleSizeR” [ 37 ].
A worthwhile alternative for planning the sample size that puts less emphasis on null hypothesis testing is based on the desired precision of the study; for example, one can calculate the sample size that is necessary to obtain a desired width of a confidence interval for the targeted effect [ 38 – 40 ]. A general framework to sample size justification beyond a calculation-only approach has been proposed [ 41 ]. It is also worth mentioning that some study types have other requirements or need specific methods. In diagnostic testing, one would need to determine the anticipated minimal sensitivity or specificity; in prognostic research, the number of parameters that can be used to fit a prediction model given a fixed sample size should be specified. Designs can also be so complex that a simulation (Monte Carlo method) may be required.
Sample size calculations should be done under different assumptions, and the largest estimated sample size is often the safer bet than a best-case scenario. The calculated sample size should further be adjusted to allow for possible missing data. Due to the complexity of accurately calculating sample size, researchers should strongly consider consulting a statistician early in the study design process.
Rule 4: Write a data management plan
In 2020, 2 Coronavirus Disease 2019 (COVID-19) papers in leading medical journals were retracted after major concerns about the data were raised [ 42 ]. Today, raw data are more often recognized as a key outcome of research along with the paper. Therefore, it is important to develop a strategy for the life cycle of data, including suitable infrastructure for long-term storage.
The data life cycle is described in a data management plan: a document that describes what data will be collected and how the data will be organized, stored, handled, and protected during and after the end of the research project. Several funders require a data management plan in grant submissions, and publishers like PLOS encourage authors to do so as well. The Wellcome Trust provides guidance in the development of a data management plan, including real examples from neuroimaging, genomics, and social sciences [ 43 ]. However, projects do not always allocate funding and resources to the actual implementation of the data management plan.
The Findable, Accessible, Interoperable, and Reusable (FAIR) data principles promote maximal use of data and enable machines to access and reuse data with minimal human intervention [ 44 ]. FAIR principles require the data to be retained, preserved, and shared preferably with an immutable unique identifier and a clear usage license. Appropriate metadata will help other researchers (or machines) to discover, process, and understand the data. However, requesting researchers to fully comply with the FAIR data principles in every detail is an ambitious goal.
Multidisciplinary data repositories that support FAIR are, for example, Dryad (datadryad.org https://datadryad.org/ ), EUDAT ( www.eudat.eu ), OSF (osf.io https://osf.io/ ), and Zenodo (zenodo.org https://zenodo.org/ ). A number of institutional and field-specific repositories may also be suitable. However, sometimes, authors may not be able to make their data publicly available for legal or ethical reasons. In such cases, a data user agreement can indicate the conditions required to access the data. Journals highlight what are acceptable and what are unacceptable data access restrictions and often require a data availability statement.
Organizing the study artifacts in a structured way greatly facilitates the reuse of data and code within and outside the lab, enhancing collaborations and maximizing the research investment. Support and courses for data management plans are sometimes available at universities. Another 10 simple rules paper for creating a good data management plan is dedicated to this topic [ 45 ].
Rule 5: Reduce bias
Bias is a distorted view in favor of or against a particular idea. In statistics, bias is a systematic deviation of a statistical estimate from the (true) quantity it estimates. Bias can invalidate our conclusions, and the more bias there is, the less valid they are. For example, in clinical studies, bias may mislead us into reaching a causal conclusion that the difference in the outcomes was due to the intervention or the exposure. This is a big concern, and, therefore, the risk of bias is assessed in clinical trials [ 46 ] as well as in observational studies [ 47 , 48 ].
There are many different forms of bias that can occur in a study, and they may overlap (e.g., allocation bias and confounding bias) [ 49 ]. Bias can occur at different stages, for example, immortal time bias in the design of the study, information bias in the execution of the study, and publication bias in the reporting of research. Understanding bias allows us researchers to remain vigilant of potential sources of bias when peer-reviewing and designing own studies. We summarized some common types of bias and some preventive steps in Table 1 , but many other forms of bias exist; for a comprehensive overview, see the Oxford University’s Catalogue of Bias [ 50 ].
| Name | Explanation | Prevention |
|---|---|---|
| Allocation bias | Systematic difference in the assignment of participants to the treatment and control group in a clinical trial. For example, the investigator knows or can predict which intervention the next eligible patient is supposed to receive due to poorly concealed randomization. | - Randomization with allocation concealment |
| Attrition bias | Attrition occurs when participants leave during a study that aims to explore the effect of continuous exposure (dropouts or withdrawal). For example, more dropouts of patients randomized to an aggressive cancer treatment. | - Good investigator–patient communication - Accessibility of clinics - Incentives to continue |
| Confounding bias | An artificial association between an exposure and an outcome because another variable is related to both the exposure and outcome. For example, lung cancer risk in coffee drinkers is evaluated, ignoring smoking status (smoking is associated with both coffee drinking and cancer). A challenge is that many confounders are unknown and/or not measured. | - Randomization (can address unmeasured confounders) When randomization is not possible: - Restriction to one level of the confounder - Matching on the levels of the confounder - Stratification and analysis within strata - Propensity score matching |
| Immortal time bias | Survival beyond a certain time point is necessary in order to be exposed (participants are “immortal” in that time period). For example, discharged patients are analyzed but were included in the treatment group only if they filled a prescription for a drug 90 days after discharge from hospital. | - Group assignment at time zero - Time-dependent analysis may be used |
| Information bias | Bias that arises from systematic differences in the collection, recall, recording, or handling of information. For example, blood pressure in the treatment arm is measured in the morning and for the control arm in the evening. | - Standardized data collection - Data collection independent from exposure or outcome (e.g., by blinding of intervention status/exposure) - Use of objective measurements |
| Publication bias | Occurs when only studies with a positive or negative result are published. Affects meta-analyses from systematic reviews and harms evidence-based medicine | - Writing a study protocol and preregistration - Publishing study protocol or registered report - Following reporting guidelines |
For a comprehensive collection, see catalogofbias.org .
Here are some noteworthy examples of study bias from the literature: An example of information bias was observed when in 1998 an alleged association between the measles, mumps, and rubella (MMR) vaccine and autism was reported. Recall bias (a subtype of information bias) emerged when parents of autistic children recalled the onset of autism after an MMR vaccination more often than parents of similar children who were diagnosed prior to the media coverage of that controversial and meanwhile retracted study [ 51 ]. A study from 2001 showed better survival for academy award-winning actors, but this was due to immortal time bias that favors the treatment or exposure group [ 52 , 53 ]. A study systematically investigated self-reports about musculoskeletal symptoms and found the presence of information bias. The reason was that participants with little computer-time overestimated, and participants with a lot of computer-time spent underestimated their computer usage [ 54 ].
Information bias can be mitigated by using objective rather than subjective measurements. Standardized operating procedures (SOP) and electronic lab notebooks additionally help to follow well-designed protocols for data collection and handling [ 55 ]. Despite the failure to mitigate bias in studies, complete descriptions of data and methods can at least allow the assessment of risk of bias.
Research execution
Rule 6: avoid questionable research practices.
Questionable research practices (QRPs) can lead to exaggerated findings and false conclusions and thus lead to irreproducible research. Often, QRPs are used with no bad intentions. This becomes evident when methods sections explicitly describe such procedures, for example, to increase the number of samples until statistical significance is reached that supports the hypothesis. Therefore, it is important that researchers know about QRPs in order to recognize and avoid them.
Several questionable QRPs have been named [ 56 , 57 ]. Among them are low statistical power, pseudoreplication, repeated inspection of data, p -hacking [ 58 ], selective reporting, and hypothesizing after the results are known (HARKing).
The first 2 QRPs, low statistical power and pseudoreplication, can be prevented by proper planning and designing of studies, including sample size calculation and appropriate statistical methodology to avoid treating data as independent when in fact they are not. Statistical power is not equal to reproducibility, but statistical power is a precondition of reproducibility as the lack thereof can result in false negative as well as false positive findings (see Rule 3 ).
In fact, a lot of QRP can be avoided with a study protocol and statistical analysis plan. Preregistration, as described in Rule 2, is considered best practice for this purpose. However, many of these issues can additionally be rooted in institutional incentives and rewards. Both funding and promotion are often tied to the quantity rather than the quality of the research output. At universities, still only few or no rewards are given for writing and registering protocols, sharing data, publishing negative findings, and conducting replication studies. Thus, a wider “culture change” is needed.
Rule 7: Be cautious with interpretations of statistical significance
It would help if more researchers were familiar with correct interpretations and possible misinterpretations of statistical tests, p -values, confidence intervals, and statistical power [ 59 , 60 ]. A statistically significant p -value does not necessarily mean that there is a clinically or biologically relevant effect. Specifically, the traditional dichotomization into statistically significant ( p < 0.05) versus statistically nonsignificant ( p ≥ 0.05) results is seldom appropriate, can lead to cherry-picking of results and may eventually corrupt science [ 61 ]. We instead recommend reporting exact p -values and interpreting them in a graded way in terms of the compatibility of the null hypothesis with the data [ 62 , 63 ]. Moreover, a p -value around 0.05 (e.g., 0.047 or 0.055) provides only little information, as is best illustrated by the associated replication power: The probability that a hypothetical replication study of the same design will lead to a statistically significant result is only 50% [ 64 ] and is even lower in the presence of publication bias and regression to the mean (the phenomenon that effect estimates in replication studies are often smaller than the estimates in the original study) [ 65 ]. Claims of novel discoveries should therefore be based on a smaller p -value threshold (e.g., p < 0.005) [ 66 ], but this really depends on the discipline (genome-wide screenings or studies in particle physics often apply much lower thresholds).
Generally, there is often too much emphasis on p -values. A statistical index such as the p -value is just the final product of an analysis, the tip of the iceberg [ 67 ]. Statistical analyses often include many complex stages, from data processing, cleaning, transformation, addressing missing data, modeling, to statistical inference. Errors and pitfalls can creep in at any stage, and even a tiny error can have a big impact on the result [ 68 ]. Also, when many hypothesis tests are conducted (multiple testing), false positive rates may need to be controlled to protect against wrong conclusions, although adjustments for multiple testing are debated [ 69 – 71 ].
Thus, a p -value alone is not a measure of how credible a scientific finding is [ 72 ]. Instead, the quality of the research must be considered, including the study design, the quality of the measurement, and the validity of the assumptions that underlie the data analysis [ 60 , 73 ]. Frameworks exist that help to systematically and transparently assess the certainty in evidence; the most established and widely used one is Grading of Recommendations, Assessment, Development and Evaluations (GRADE; www.gradeworkinggroup.org ) [ 74 ].
Training in basic statistics, statistical programming, and reproducible analyses and better involvement of data professionals in academia is necessary. University departments sometimes have statisticians that can support researchers. Importantly, statisticians need to be involved early in the process and on an equal footing and not just at the end of a project to perform the final data analysis.
Rule 8: Make your research open
In reality, science often lacks transparency. Open science makes the process of producing evidence and claims transparent and accessible to others [ 75 ]. Several universities and research funders have already implemented open science roadmaps to advocate free and public science as well as open access to scientific knowledge, with the aim of further developing the credibility of research. Open research allows more eyes to see it and critique it, a principle similar to the “Linus’s law” in software development, which says that if there are enough people to test a software, most bugs will be discovered.
As science often progresses incrementally, writing and sharing a study protocol and making data and methods readily available is crucial to facilitate knowledge building. The Open Science Framework (osf.io) is a free and open-source project management tool that supports researchers throughout the entire project life cycle. OSF enables preregistration of study protocols and sharing of documents, data, analysis code, supplementary materials, and preprints.
To facilitate reproducibility, a research paper can link to data and analysis code deposited on OSF. Computational notebooks are now readily available that unite data processing, data transformations, statistical analyses, figures and tables in a single document (e.g., R Markdown, Jupyter); see also the 10 simple rules for reproducible computational research [ 76 ]. Making both data and code open thus minimizes waste of funding resources and accelerates science.
Open science can also advance researchers’ careers, especially for early-career researchers. The increased visibility, retrievability, and citations of datasets can all help with career building [ 77 ]. Therefore, institutions should provide necessary training, and hiring committees and journals should align their core values with open science, to attract researchers who aim for transparent and credible research [ 78 ].
Research reporting
Rule 9: report all findings.
Publication bias occurs when the outcome of a study influences the decision whether to publish it. Researchers, reviewers, and publishers often find nonsignificant study results not interesting or worth publishing. As a consequence, outcomes and analyses are only selectively reported in the literature [ 79 ], also known as the file drawer effect [ 80 ].
The extent of publication bias in the literature is illustrated by the overwhelming frequency of statistically significant findings [ 81 ]. A study extracted p -values from MEDLINE and PubMed Central and showed that 96% of the records reported at least 1 statistically significant p -value [ 82 ], which seems implausible in the real world. Another study plotted the distribution of more than 1 million z -values from Medline, revealing a huge gap from −2 to 2 [ 83 ]. Positive studies (i.e., statistically significant, perceived as striking or showing a beneficial effect) were 4 times more likely to get published than negative studies [ 84 ].
Often a statistically nonsignificant result is interpreted as a “null” finding. But a nonsignificant finding does not necessarily mean a null effect; absence of evidence is not evidence of absence [ 85 ]. An individual study may be underpowered, resulting in a nonsignificant finding, but the cumulative evidence from multiple studies may indeed provide sufficient evidence in a meta-analysis. Another argument is that a confidence interval that contains the null value often also contains non-null values that may be of high practical importance. Only if all the values inside the interval are deemed unimportant from a practical perspective, then it may be fair to describe a result as a null finding [ 61 ]. We should thus never report “no difference” or “no association” just because a p -value is larger than 0.05 or, equivalently, because a confidence interval includes the “null” [ 61 ].
On the other hand, studies sometimes report statistically nonsignificant results with “spin” to claim that the experimental treatment is beneficial, often by focusing their conclusions on statistically significant differences on secondary outcomes despite a statistically nonsignificant difference for the primary outcome [ 86 , 87 ].
Findings that are not being published have a tremendous impact on the research ecosystem, distorting our knowledge of the scientific landscape by perpetuating misconceptions, and jeopardizing judgment of researchers and the public trust in science. In clinical research, publication bias can mislead care decisions and harm patients, for example, when treatments appear useful despite only minimal or even absent benefits reported in studies that were not published and thus are unknown to physicians [ 88 ]. Moreover, publication bias also directly affects the formulation and proliferation of scientific theories, which are taught to students and early-career researchers, thereby perpetuating biased research from the core. It has been shown in modeling studies that unless a sufficient proportion of negative studies are published, a false claim can become an accepted fact [ 89 ] and the false positive rates influence trustworthiness in a given field [ 90 ].
In sum, negative findings are undervalued. They need to be more consistently reported at the study level or be systematically investigated at the systematic review level. Researchers have their share of responsibilities, but there is clearly a lack of incentives from promotion and tenure committees, journals, and funders.
Rule 10: Follow reporting guidelines
Study reports need to faithfully describe the aim of the study and what was done, including potential deviations from the original protocol, as well as what was found. Yet, there is ample evidence of discrepancies between protocols and research reports, and of insufficient quality of reporting [ 79 , 91 – 95 ]. Reporting deficiencies threaten our ability to clearly communicate findings, replicate studies, make informed decisions, and build on existing evidence, wasting time and resources invested in the research [ 96 ].
Reporting guidelines aim to provide the minimum information needed on key design features and analysis decisions, ensuring that findings can be adequately used and studies replicated. In 2008, the Enhancing the QUAlity and Transparency Of Health Research (EQUATOR) network was initiated to provide reporting guidelines for a variety of study designs along with guidelines for education and training on how to enhance quality and transparency of health research. Currently, there are 468 reporting guidelines listed in the network; see the most prominent guidelines in Table 2 . Furthermore, following the ICMJE recommendations, medical journals are increasingly endorsing reporting guidelines [ 97 ], in some cases making it mandatory to submit the appropriate reporting checklist along with the manuscript.
| Guideline name | Study type |
|---|---|
| ARRIVE | Animal experiments |
| CONSORT | Randomized trials |
| STROBE | Observational studies |
| PRISMA | Systematic reviews |
| SPIRIT | Study protocols |
| STARD/TRIPOID | Diagnostic/prognostic studies |
The EQUATOR Network is a library with more than 400 reporting guidelines in health research ( www.equator-network.org ).
The use of reporting guidelines and journal endorsement has led to a positive impact on the quality and transparency of research reporting, but improvement is still needed to maximize the value of research [ 98 , 99 ].
Conclusions
Originally, this paper targeted early-career researchers; however, throughout the development of the rules, it became clear that the present recommendations can serve all researchers irrespective of their seniority. We focused on practical guidelines for planning, conducting, and reporting of research. Others have aligned GRP with similar topics [ 100 , 101 ]. Even though we provide 10 simple rules, the word “simple” should not be taken lightly. Putting the rules into practice usually requires effort and time, especially at the beginning of a research project. However, time can also be redeemed, for example, when certain choices can be justified to reviewers by providing a study protocol or when data can be quickly reanalyzed by using computational notebooks and dynamic reports.
Researchers have field-specific research skills, but sometimes are not aware of best practices in other fields that can be useful. Universities should offer cross-disciplinary GRP courses across faculties to train the next generation of scientists. Such courses are an important building block to improve the reproducibility of science.
Acknowledgments
This article was written along the Good Research Practice (GRP) courses at the University of Zurich provided by the Center of Reproducible Science ( www.crs.uzh.ch ). All materials from the course are available at https://osf.io/t9rqm/ . We appreciated the discussion, development, and refinement of this article within the working group “training” of the SwissRN ( www.swissrn.org ). We are grateful to Philip Bourne for a lot of valuable comments on the earlier versions of the manuscript.
Funding Statement
S.S. received funding from SfwF (Stiftung für wissenschaftliche Forschung an der Universität Zürich; grant no. STWF-19-007). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.

IMAGES
VIDEO
COMMENTS
Learn a step-by-step method for turning research results into a published paper using the LEAP writing approach. The LEAP approach helps you first think about and then write each section of the research paper, from laying out the facts to preparing for submission.
Rule 1: Make It a Driving Force. Never separate writing a paper from the underlying research. After all, writing and research are integral parts of the overall enterprise. Therefore, design a project with an ultimate paper firmly in mind.
Learn the steps to write a research paper, from choosing a topic to proofreading your draft. This guide covers the basics of academic writing, research, and argumentation with examples and tips.
Learn how to write a research paper properly with this concise guide that covers topics like choosing a topic, gathering sources, writing a thesis, and citing evidence. Find out the difference between a research paper and a research proposal, and get tips on formatting, length, and style.
Learn how to write a research paper with this comprehensive guide that covers the definition, structure, methods, and examples of a research paper. Find out how to choose a topic, conduct a literature review, develop a thesis, collect and analyze data, organize your paper, and cite your sources.
Learn how to choose a topic, conduct research, define a question, write a thesis, and structure your paper with this comprehensive guide. Find tips on formatting, citation, and ethical norms for your research paper.
Learn how to format your research paper according to APA, MLA, or Chicago style. Download free templates and get tips on title page, headings, citations, and more.
Research Paper Structure: A Snapshot. Before diving into the individual components, let's take a quick look at the full structure of a research paper. This snapshot will help you visualize how each section fits together to form a cohesive and well-organized paper. Introduction. Introduce your topic and research question.
Learn how to write an effective introduction for a research paper, whether it's argumentative or empirical. Follow the five steps to introduce your topic, provide background, establish your problem, specify your objective, and map out your paper.
One of the stumbling blocks is the beginning of the process and creating the first draft. This paper presents guidelines on how to initiate the writing process and draft each section of a research manuscript. The paper discusses seven rules that allow the writer to prepare a well-structured and comprehensive manuscript for a publication submission.
Learn how to choose a topic, plan, research, write, and edit a research paper with this comprehensive guide. Find out the difference between report and thesis papers, the reasons for writing a research paper, and the tips and tricks to make your paper stand out.
Learn the stages and skills involved in writing a library-based research paper, from finding and selecting sources to revising and documenting. This web page also provides links to other resources for academic and professional writing.
Learn how to choose a topic, do research, write a thesis statement, create an outline, and structure your paper with this comprehensive guide. Find tips and examples for academic writing that involves information analysis, source evaluation, and argument explanation.
These rules are designed to make your paper more influential and the process of writing more efficient and pleasurable. Introduction. Writing and reading papers are key skills for scientists. ... It is also the bridge that connects the experimental phase of a research effort with the paper-writing phase. Thus, it is useful to formalize the ...
Learn how to choose a topic, find sources, organize your notes, write a draft, and revise your paper with this guide from the Writing Center at Potsdam. Follow the steps to understand the assignment, set a schedule, begin research, construct an outline, write a draft, and write a final draft.
Learn how to write a research paper with this comprehensive guide from Purdue OWL. Find out how to choose a topic, identify an audience, and follow the steps of the writing process.
Learn how to create and format APA Style student papers with this step-by-step guide. See annotated diagrams, sample papers, and tips for title page, text, tables, figures, and reference list.
Learn how to write a research paper for a peer-reviewed journal, with tips on structure, content, and style. A research paper is a highly codified rhetorical form that addresses a specific research question and follows the IMRAD format: Introduction, Methods, Results, and Discussion.
Learn how to choose a topic, find sources, make an outline, and write a research paper with this helpful guide. Get tips on citation, analysis, discussion, and conclusion, and use free online tools for assistance.
This article offers advice on how to write and publish papers in computational biology, covering topics such as project design, audience, logic, completeness, and self-containment. It is not about the mechanics of composing a paper, but about the principles and attitude that can help guide the process of writing and research.
Learn the basics of writing a research paper for an academic course or journal, including how to choose a topic, conduct research, determine a thesis and build an outline. Follow the steps and tips to create a strong and informative paper that meets the guidelines and rules of your assignment.
Properly Quoting And Paraphrasing. When you use someone else's ideas or words, you must cite them. Here are some tips: Quotation: Use the exact words from a source, placing them in quotation marks and citing the source.; Paraphrasing: Rewrite the idea in your own words while still giving credit to the original author.; Summarizing: Condense the main ideas of a source into a brief overview, and ...
validity of the results during the research. As a result, the overall research may need. to be adjusted, the project design may be. revised, new methods may be devised, and. new data may be ...
Learn how to write a scientific article for publication in a medical journal, from the background work to the final submission. This guide covers the main sections, elements and tips for each section, with examples and references.
Rule 4: Write a data management plan. In 2020, 2 Coronavirus Disease 2019 (COVID-19) papers in leading medical journals were retracted after major concerns about the data were raised [42]. Today, raw data are more often recognized as a key outcome of research along with the paper.